Medical device and MedTech insights, news, tips and more
FDA Clears Omega3-Rich Fish Skin for Surgical Applications
September 16, 2016
MCLEAN, VA–(Marketwired – Sep 13, 2016) – Kerecis, the company using Omega3-rich fish skin for tissue healing, has received clearance from the Food and Drug Administration (FDA) to market the proprietary Kerecis technology as a surgical buttress in the United States. The Kerecis Omega3 SecureMesh can be used in lung, bariatric, gastric, colorectal and other surgeries.
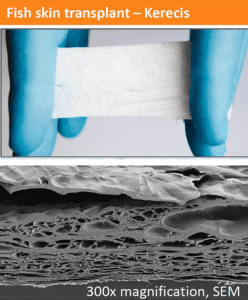 Kerecis Omega3 is intact fish skin rich in naturally occurring Omega3 polyunsaturated fatty acids. The material, which is used to regenerate damaged human tissue, has the potential to accelerate healing, and was developed in collaboration with the U.S. Office of Naval Research.
Kerecis Omega3 is intact fish skin rich in naturally occurring Omega3 polyunsaturated fatty acids. The material, which is used to regenerate damaged human tissue, has the potential to accelerate healing, and was developed in collaboration with the U.S. Office of Naval Research.
Surgical staplers are commonly used during laparoscopic operations to simultaneously cut organs and staple the resulting organ wound closed. The Kerecis Omega3 fish-skin product can strengthen the staple line and reduce the possibility of leakage. The Kerecis Omega3 material recruits the body’s cells from the tissue surrounding the organ cut. These cells are incorporated into the fish skin, which is ultimately converted into functional, living tissue. Studies have shown that cells and stem cells proliferate faster in this structure than in mammalian-sourced materials such as pericardium tissue.
“With diabetes and obesity so common today, improved surgical technologies are needed to cope with more stress and tissue mass,” said Fertram Sigurjonsson, CEO, president and chairman of Kerecis Limited. “Our technology can help improve outcomes by reducing the possibility of leakage from surgical wounds. The FDA approval represents an important milestone for us, as we now have two approved products in the world’s largest market for surgical products.”
About Kerecis
Kerecis develops regenerative technologies based on fish skin and Omega3 polyunsaturated fatty acids.
The Kerecis technology is patented in the U.S. and multiple other countries. The initial Kerecis product, Kerecis Omega3 Wound, has been approved by the FDA and European regulatory authorities for wound healing. The product has a Medicare “Q” code (Q4158), allowing it to be easily identified and processed by Medicare and private insurance companies. The FDA has also cleared Kerecis Omega3 SecureMesh for surgical use in the United States.
Kerecis Omega3 is produced in Iceland from fish locally harvested in the North Atlantic waters. For more information, visit www.kerecis.com. Distributor and licensing inquiries are welcome.
Source: FDA Clears Omega3-Rich Fish Skin for Surgical Applications
