Medical device and MedTech insights, news, tips and more
7D Surgical Receives FDA 510(K) and Health Canada MDL Clearance for Its Breakthrough Image Guidance
February 1, 2017
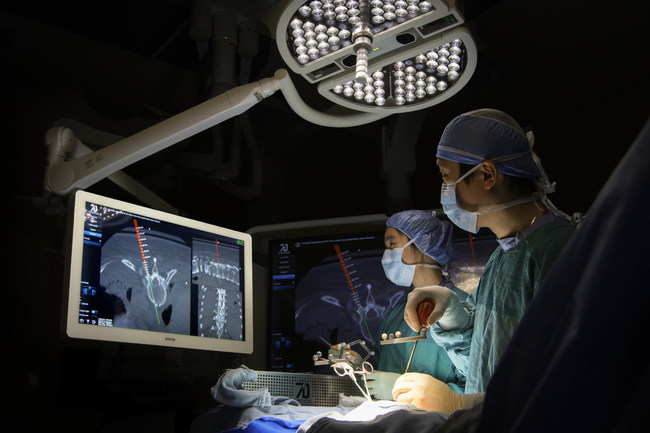
TORONTO, Jan. 23, 2017 /PRNewswire/ — 7D Surgical announced today that it has received both 510(k) clearance from the U.S. Food and Drug Administration (FDA) and a medical device license from Health Canada enabling the North American commercial launch of its innovative Machine-vision Image Guided Surgery (MIGS™) system for spine surgery, the 7D Surgical System.
The 7D Surgical System employs cutting-edge 3D optical technologies and machine vision algorithms to eliminate the long-standing barriers to adoption of existing surgical navigational platforms. This new technology can easily register spinal surgery patients automatically using only visible light. Unlike time-consuming conventional image guided surgery (IGS) systems that depend on intraoperative radiation, this new platform can achieve an incredibly fast surgical workflow for spine procedures.

“When navigating the spine, surgeons traditionally have had two time-consuming and expensive IGS options: systems that rely on intraoperative radiation emitting devices or systems that utilize laborious manual point matching techniques,” said Beau Standish, Chief Executive Officer of 7D Surgical. “We believe the inefficiencies of these systems have limited the adoption of IGS in spine procedures to less than 20%. 7D Surgical’s MIGS™ system has now removed these barriers, providing surgeons and their hospitals with a superior product option.”
The 7D Surgical System enables near-instantaneous Flash Registration™ of the patient’s anatomy. “Guided by our product philosophy of ‘surgeons designing for surgeons’, we have achieved an unprecedented entire workflow time of less than 20 seconds for de novo spinal registration, unheard of in the spinal IGS world where such registration can interrupt surgery for up to 30 minutes,” said Dr. Victor Yang, President and Chief Scientific Officer of 7D Surgical, Senior Scientist at Sunnybrook Research Institute, and Staff Neurosurgeon at Sunnybrook Health Sciences Centre in Toronto, where prototype MIGS™ technology has been used in more than 160 patients in clinical trials.
Read More – Source: 7D Surgical Receives FDA 510(K) and Health Canada MDL Clearance for Its Breakthrough Image Guidance
