Medical device and MedTech insights, news, tips and more
Advanced Sterilization Products Unveils the Fastest Biological Indicator for Hydrogen Peroxide Sterilization
January 28, 2020
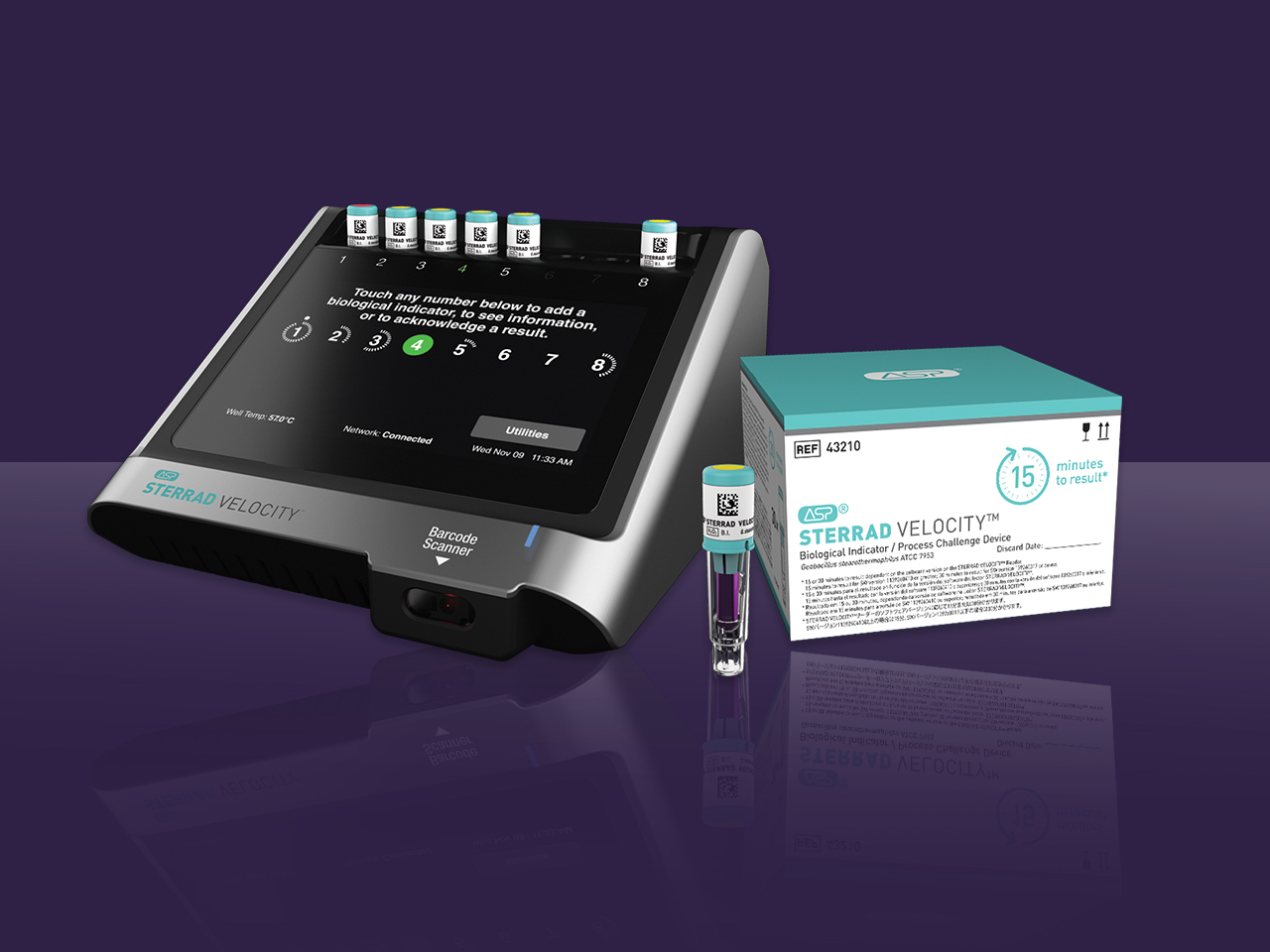
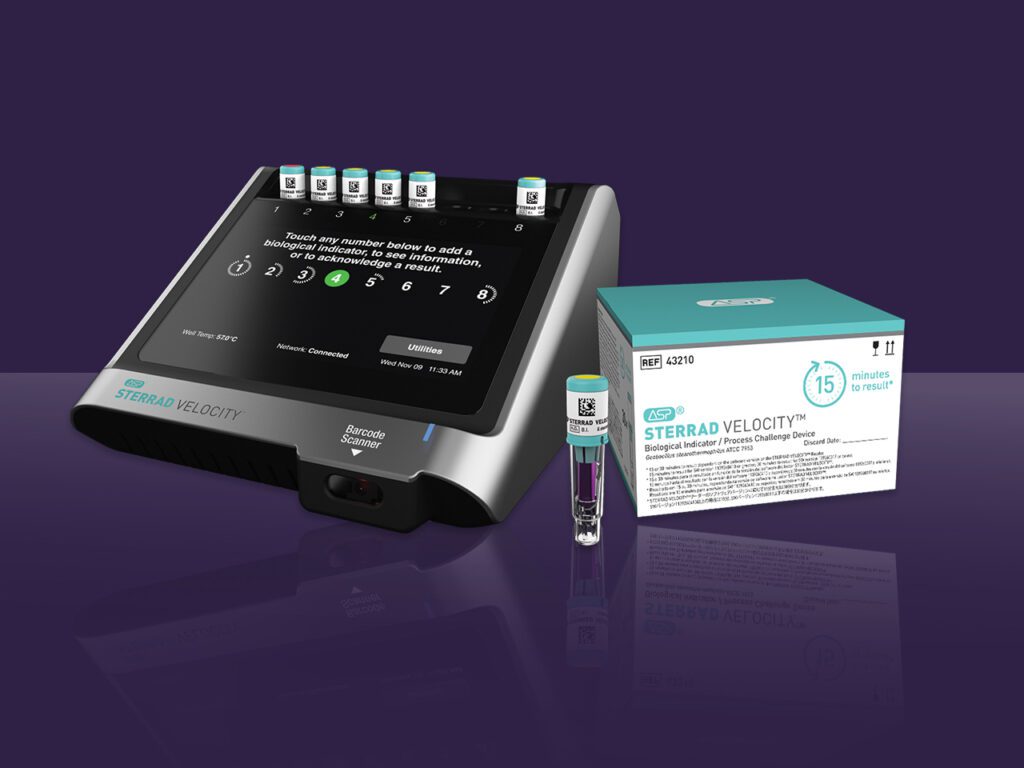
Advanced Sterilization Products (ASP) announced today it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for a 15-minute time to result on the STERRAD VELOCITY® Biological Indicator (BI)/Process Challenge Device (PCD) for use in STERRAD® Systems.* The 15-minute time to result is up to 38% faster than competition and offers the fastest way to provide instrument sterility assurance for healthcare professionals.
“Every minute matters in sterile processing,” notes Amy Smith, VP Global Marketing for ASP. “ASP Innovation to cut the BI read time in half allows customers to assure sterility for every instrument per AAMI guidelines while keeping up with the pace of a busy SPD. This is just another way ASP continues to partner with the healthcare community to elevate the standard of care by protecting patients at their most critical moments.”
STERRAD VELOCITY® BI/PCD is used to monitor loads and assess process performance for vaporized hydrogen peroxide (H2O2) sterilization. This is a crucial step to verify load sterility for critical and semi-critical surgical instruments prior to releasing them to the operating room. AAMI ST58:2013/(R) 2018 states frequent monitoring may mitigate the many factors that could affect cycle variation and reduce risk to patients. It is for this reason AAMI ST58 recommends using a PCD with the appropriate BI daily, but preferably in every sterilization cycle. STERRAD VELOCITY® BI/PCD is the only rapid-read PCD validated for STERRAD® Systems that meets AAMI guidelines.
STERRAD VELOCITY® BI/PCD is commercially available in the United States. Existing STERRAD VELOCITY® System customers should contact ASP to get the 15-minute result with a software upgrade to their reader.
ABOUT ADVANCED STERILIZATION PRODUCTS (ASP)
Advanced Sterilization Products (ASP) has a long track record of designing and delivering innovative infection prevention solutions that dramatically raise the level of health care and safety for those who matter most. Our pioneering technology, global distribution and established leadership position enable us to simplify the process of buying and operating infection prevention products and services every day for thousands of medical facilities around the world. This enables our customers to focus on what they do best – preventing infection and saving lives. For more information, please visit www.ASP.com.
See Full Press Release: Advanced Sterilization Products Unveils the Fastest Biological Indicator for Hydrogen Peroxide Sterilization, Available Exclusively With the STERRAD VELOCITY® System | Business Wire
Written by: Advanced Sterilization Products (ASP)

Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.