Medical device and MedTech insights, news, tips and more
Ancora Heart’s AccuCinch System Demonstrates Significant Improvement in Quality of Life, Cardiac Structure and Function in Heart Failure Patients
March 24, 2023
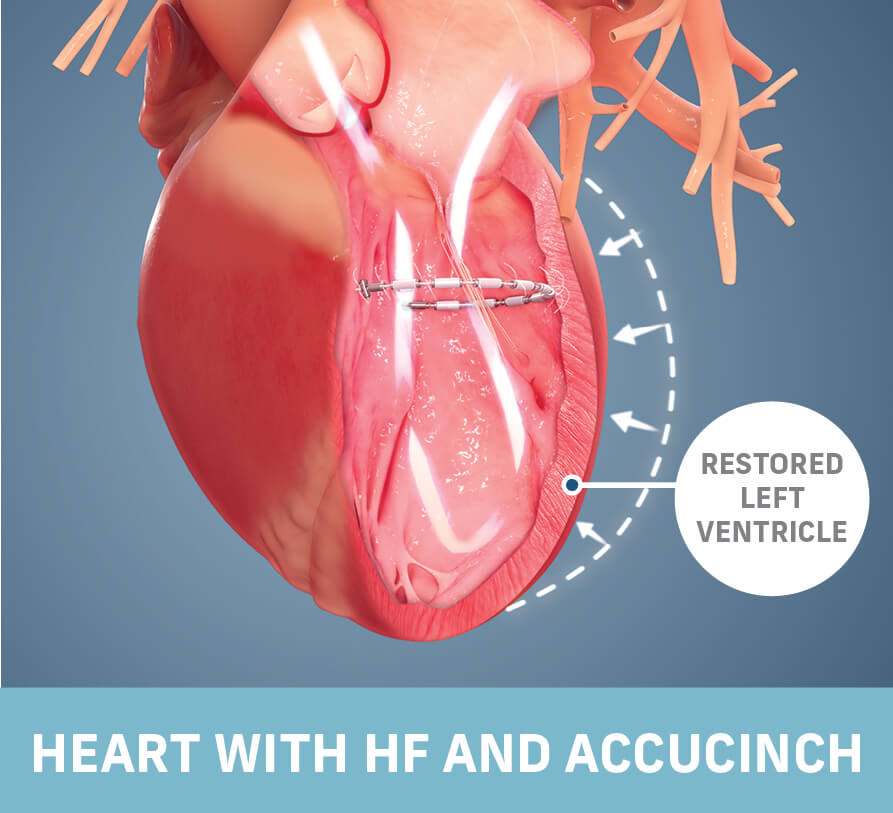
Ancora Heart, Inc., a company developing a completely transcatheter device-based therapy to address heart failure (HF), announced that patients treated with the investigational AccuCinch® Ventricular Restoration System demonstrated improvement in HF patient outcomes and beneficial changes in the structure of the heart. The 12-month data were presented as part of a late-breaking clinical science session at the Technology and Heart Failure Therapeutics conference (THT 2023) and simultaneously published in the Journal of Cardiac Failure.
The presentation, titled “Transcatheter Left Ventricular Restoration in Patients with Heart Failure” was presented by Nadira Hamid, MD, interventional echocardiographer, cardiologist at Allina Health Minneapolis Heart Institute and assistant director of the echocardiographic core lab at CRF, on Tuesday, March 21. Dr. Hamid is also co-author of the corresponding manuscript, titled: “Transcatheter Left Ventricular Restoration in Patients with Heart Failure.”
The data will include complete 12-month clinical outcomes from an analysis of 51 symptomatic heart failure patients with reduced ejection fraction (HFrEF) who were treated with the AccuCinch System. Patients included in the analysis matched key inclusion criteria (LVEF 20-40%, MR ≤ 2+) of the currently enrolling CORCINCH-HF pivotal trial.
“This analysis shows patients treated with the AccuCinch System experienced meaningful improvement in efficacy measures important to both clinicians and patients,” said Dr. Hamid. “We witnessed significant and progressive reverse remodeling of the left ventricle, as well as significant and sustained improvement in quality of life and functional capacity measures. These results show promise that this innovative catheter-based therapy may help broaden options for treating patients with heart failure, and we look forward to results of the pivotal study that’s underway.”
The primary outcome measure for this analysis was the change of left ventricular end-diastolic volume (LVEDV) compared to baseline through 12 months. Other efficacy assessments included changes from baseline in Kansas City Cardiomyopathy Questionnaire (KCCQ) scores, six-minute walk test (6MWT) distances, New York Heart Association (NYHA) classification and ejection fraction (EF). The safety outcome measure included freedom from major adverse events (MAEs) related to the device implantation or femoral artery access through 12 months post-procedure.
Key findings include:
- Significant and progressive improvement in left ventricular end diastolic volume (LVEDV) at 12 months compared to baseline (-33.6 ± 34.8 ml, p<0.001),
- Significant and sustained improvement in overall quality of life scores (KCCQ-OS) at 12 months compared to baseline (16.4 ± 18.7 points, p<0.001)
- Significant and sustained improvement in functional capacity (6MWT distances) at 12 months compared to baseline (45.9 ± 83.9 m, p<0.001)
- Significant and sustained improvement in New York Heart Association (NYHA) classification at 12 months (p<0.001) compared to baseline (improved ≥ 1 Class: 65%; improved or unchanged: 94%)
- Significant and progressive improvement in ejection fraction (EF) at 12 months compared to baseline (3.1 ± 6.9, p<0.001)
“Heart failure is a very prevalent disease, and because of its progressive nature, medications and device treatments may become less effective over time, leaving patients with persistent symptoms and limited treatment options,” said Ulrich Jorde, MD, professor of medicine, Albert Einstein College of Medicine, and section head of Heart Failure, Cardiac Transplantation and Mechanical Circulatory Support at Montefiore Health System in New York. “Utilizing a fully transcatheter approach to directly treat a structural driver of heart failure progression, the enlarged left ventricle, we believe there may be a new opportunity to improve left ventricular end diastolic volume and help patients feel better and this 12-month AccuCinch System data clearly demonstrate its potential to address this important unmet need.”1
The pivotal CORCINCH-HF Study, a randomized, controlled trial, is currently underway to assess the clinical safety and efficacy of the AccuCinch System in HFrEF patients.
“The AccuCinch procedure is designed to help us achieve predictable and repeatable reduction in LV dimensions,” said Mark Reisman, MD, director of structural heart disease at NewYork-Presbyterian/Weill Cornell Medical Center and Weill Cornell Medicine, and co-director of structural heart disease for NewYork-Presbyterian Queens and NewYork-Presbyterian Brooklyn Methodist Hospital. “We look forward to seeing the accelerated cadence of enrollment in the trial and assessing the impact on our patients.”
“The AccuCinch System continues to demonstrate tremendous potential to fill a need in heart failure treatment options, and today’s analysis shows its ability to improve the health of the left ventricle in a safe and effective manner,” said Jeff Closs, president and CEO of Ancora Heart. “We are excited about the significant and sustained results of this 12-month analysis and look forward to completion of the CORCINCH-HF pivotal trial and eventual submission to the FDA.”
An international heart failure conference organized by the Cardiovascular Research Foundation (CRF), THT 2023 is an immersive three-day experience covering the field’s cutting-edge technology and devices, along with innovations in the pipeline.
About Ancora Heart
Ancora Heart is a medical device company dedicated to providing new treatment options for people with heart failure (HF). The company’s proprietary AccuCinch® Ventricular Restoration System is the only completely transcatheter device designed to restore the structure and function of the enlarged left ventricle of the heart, thereby addressing the fundamental issue in the progression of heart failure in patients with reduced ejection fraction (HFrEF).. Ancora Heart is a privately held company located in Santa Clara, Calif. For more information, please visit www.ancoraheart.com and connect on Twitter (@AncoraHeart), LinkedIn and Facebook.
See Full Press Release at the Source: Ancora Heart’s AccuCinch System Demonstrates Significant Improvement in Quality of Life, Cardiac Structure and Function in Heart Failure Patients
Press Release by: Ancora Heart
Legacy MedSearch has more than 35 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 17 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.

