Medical device and MedTech insights, news, tips and more
Auris Health’s Lung Cancer Detecting Robot has Late-Breaking Results from its First-in-Human Study
October 23, 2019
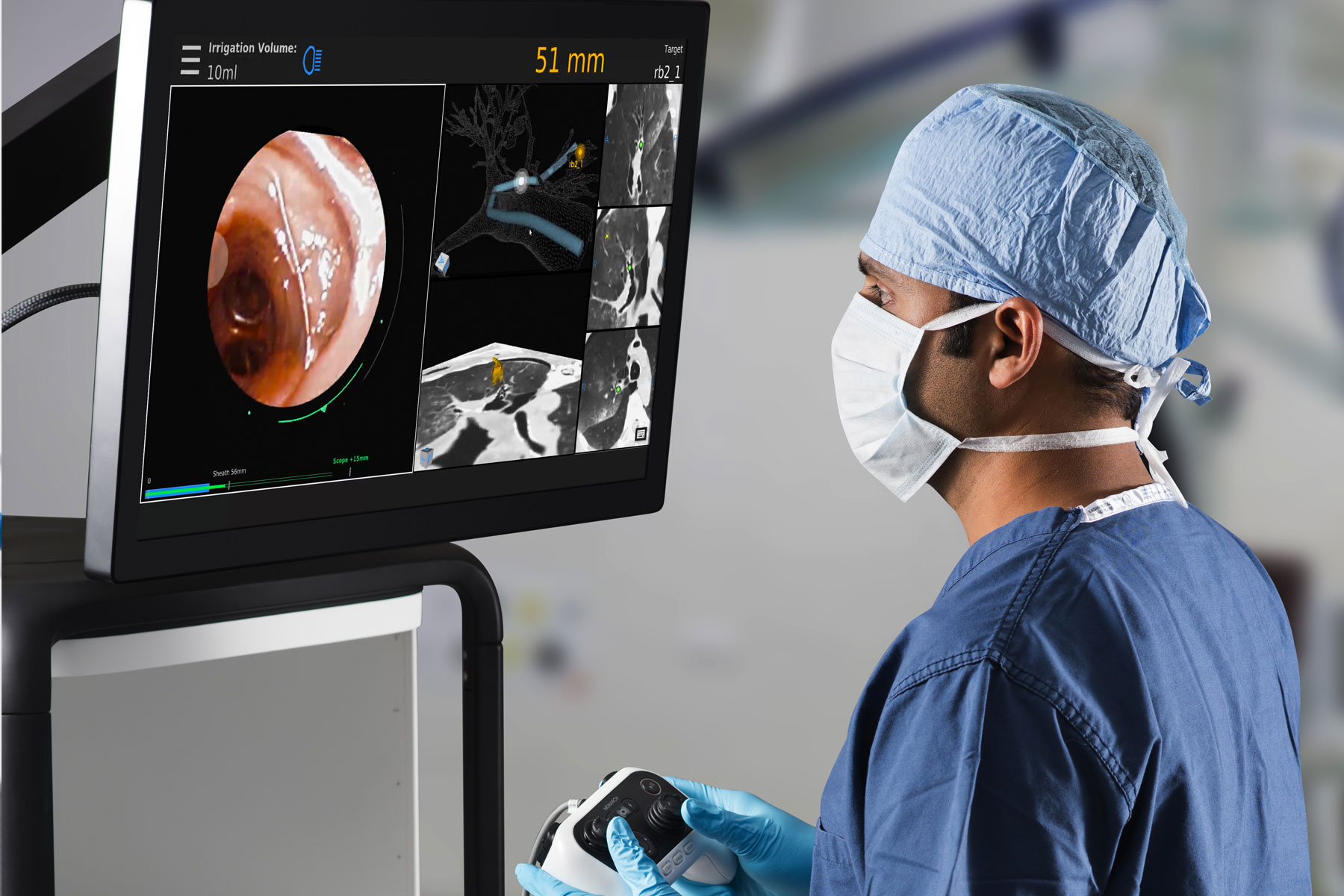
Auris Health, Inc., part of Johnson & Johnson Medical Devices Companies,* announced results from a study demonstrating the safety and feasibility of the Monarch Platform, a first-of-its-kind robotic technology indicated for use in diagnostic and therapeutic bronchoscopic procedures. Data from the BENEFIT study was presented in a late-breaking session at the CHEST 2019 Conference in New Orleans by Alexander C. Chen, M.D., of St. Louis, Mo.
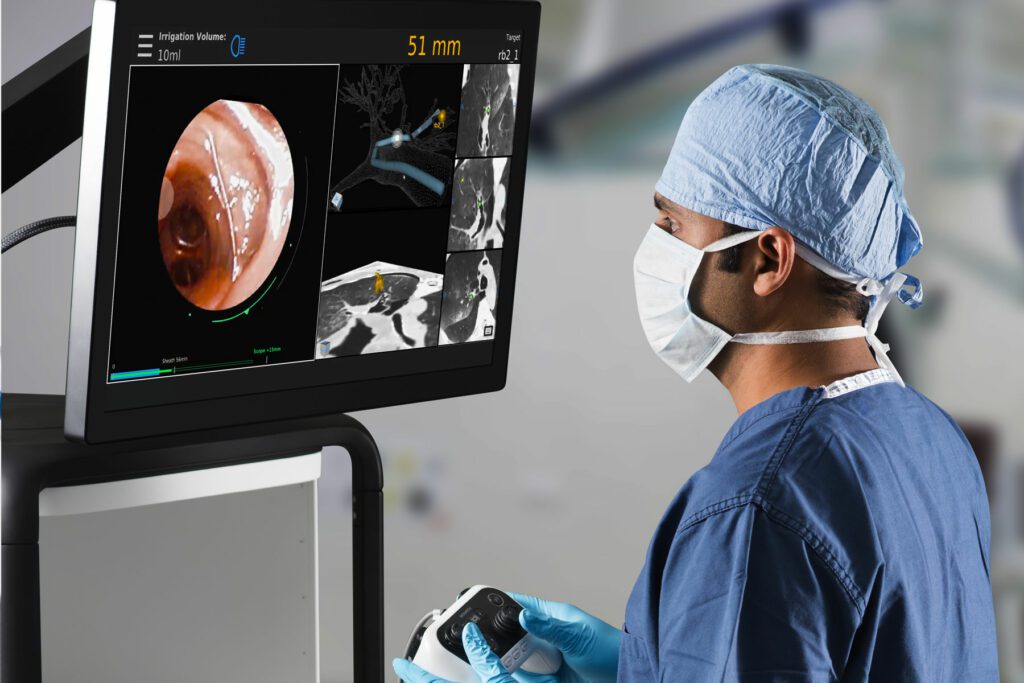
The BENEFIT study assessed the safety and feasibility of utilizing a robotic system to aid in the diagnosis of peripheral pulmonary lesions 1-5 cm in size. During the study, investigators performed robotic-assisted bronchoscopy using the Monarch Platform on 55 patients across five study sites. The Monarch Platform localized targeted nodules using a combination of direct visualization, navigational guidance and radial endobronchial ultrasound (R-EBUS). The primary effectiveness endpoint, successful localization of targeted pulmonary lesions, was reported in 52/54 (96.3%) of procedures. Pneumothorax occurred in 2/55 (3.6%) of procedures and required tube thoracostomy in 1/55 (1.8%) procedure. No additional serious adverse events were reported.
“This is the first prospective, multicenter study of robot-assisted bronchoscopy in patients with undiagnosed lung lesions,” said Gerard A. Silvestri, Hillenbrand Professor of Thoracic Oncology and a lung cancer pulmonologist at the Medical University of South Carolina. “We were able to safely perform the procedure and successfully localize lesions in 96% of cases. This data suggests robot-assisted bronchoscopy is feasible and may improve bronchoscopists’ ability to effectively localize peripheral lesions. The promising results of this study warrant further evaluation in additional prospective studies where yield can be better assessed.”
“The results from the BENEFIT study demonstrate the potential of Monarch to help physicians diagnose these hard-to-reach lesions,” said Josh DeFonzo, Chief Operating Officer of Auris Health. “We are very encouraged by the results of the BENEFIT study and are thrilled to be making a positive impact on the diagnosis of lung conditions. We will continue to build robust clinical evidence around the Monarch Platform to support its use in interventional pulmonology.”
Since Auris Health commercially launched the Monarch Platform last year, more than 1,000 procedures have been performed using the technology in the United States.
See the Full Press Release: Auris Health Announces Late-Breaking Results from First-in-Human Study of Monarch™ Platform
Written by: Auris Health

Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.
