Medical device and MedTech insights, news, tips and more
Cardiovalve Wins FDA Breakthrough Device Designation for Transcatheter Tricuspid Valve Replacement System
March 5, 2020
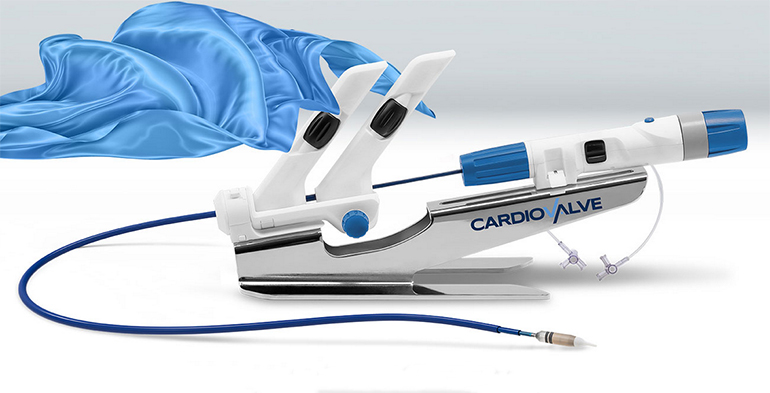
Cardiovalve announced that it has received U.S. Food and Drug Administration (FDA) approval for an Early Feasibility Study (EFS) of its Transcatheter Tricuspid Valve Replacement System for a tricuspid regurgitation (TR) indication. The Cardiovalve System also has been granted ‘Breakthrough Device Designation’ status by the FDA.
Cardiovalve is the first privately held company to have the FDA’s EFS approval for both TR and MR (tricuspid/mitral regurgitation) indications.
“I am excited to finally have a truly percutaneous approach for treating TR. The Cardiovalve System is an innovative solution for an unmet clinical need,” said Dr. Azeem Latib, Medical Director of Structural Heart Interventions at Montefiore Medical Center’s Department of Cardiology in New York City. As primary investigator for the U.S. TR study, Dr. Latib will be presenting details of Cardiovalve’s technology and procedure, as well as the clinical EFS design, at the upcoming ‘CRT2020’ scientific conference on Sunday, Feb. 23, at 3:00 p.m. EST in room ‘Potomac C’ at Gaylord National Convention Center, National Harbor, Md.
Amir Gross, CEO of Cardiovalve, added: “We are excited about the FDA’s recognition of the potential clinical benefit of the Transcatheter Tricuspid Valve Replacement System. We now have clinical data two years out that our implant is functioning as expected and the patient is improved clinically. FDA’s ‘breakthrough’ designation validates and reaffirms our commitment to improving and extending the lives of millions of patients suffering from heart valve disease across the world.”
Cardiovalve commenced its Early Feasibility Study for TR in the U.S. earlier this year. The study’s primary endpoint is the safety and feasibility of the Cardiovalve technology and procedure in reducing tricuspid regurgitation, with evaluations at 30 days and periodically up to 5 years. The study will be conducted in collaboration with five leading US hospitals.
Cardiovalve has developed an innovative transfemoral valve replacement system where the valve mimics current surgical replacement solutions that can fit both mitral and tricuspid indications, covering 90% of the patient population with three approved valve sizes, using the same delivery system.
“I have previously successfully performed initial First-in-Human cases with the Cardiovalve MR procedure. Now, the Cardiovalve system can be utilized in the Tricuspid location with the same transcatheter procedure. In fact, the Tricuspid is simpler and faster and, therefore, I anticipate similar procedural success,” explained Prof. Francesco Maisano, Head of Cardiovascular Surgery at Zurich University Hospital, Switzerland.
See Full Press Release: Cardiovalve Wins FDA Breakthrough Device Designation for Transcatheter Tricuspid Valve Replacement System | Medgadget
Written by: Cardiovalve

Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.

