Medical device and MedTech insights, news, tips and more
CLEW Medical Receives FDA Clearance for AI-Based Software that Predicts Hemodynamic Instability
February 7, 2021
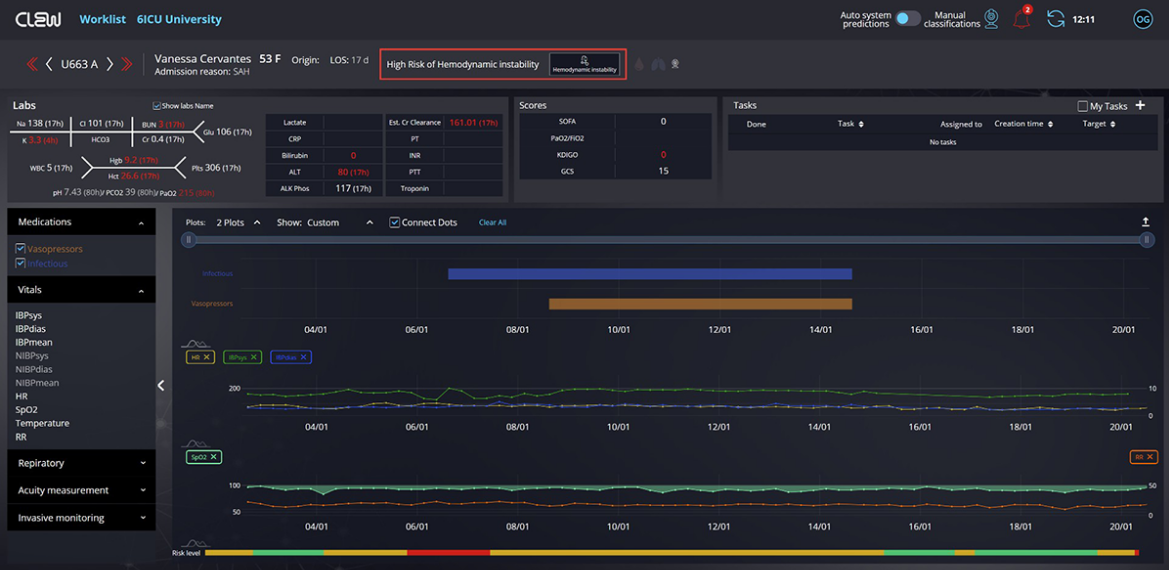
CLEW Medical, a leader in AI-powered predictive analytics, today announced that the U.S. Food and Drug Administration (FDA) has given 510(k) clearance and authorized the use of “CLEWICU,” CLEW’s artificial intelligence (AI) based ICU solution, to predict hemodynamic instability in adult patients. The clearance is the FDA’s first for such a device, and follows the FDA’s Emergency Use Authorization (EUA) for CLEWICU’s respiratory deterioration model granted in June 2020, for the predictive screening of COVID-19 and other ICU patients.
The COVID-19 pandemic has underscored the critical need for quick and accurate decision-making in intensive care units, as ICU capacity has faced unprecedented volumes. Conducting effective risk evaluation to improve patient identification and subsequent care plans requires the use of advanced tools that can provide comprehensive, predictive data to help medical professionals identify patients whose health conditions are likely to deteriorate, in addition to patients whose conditions are unlikely to deteriorate.
CLEWICU continuously monitors and categorizes patient risk levels, providing clinicians with physiological insight into a patient’s likelihood of future hemodynamic instability. The system provides notification of clinical deterioration up to eight hours in advance, enabling early evaluation and subsequent intervention for prompt, proactive patient care. The system also identifies low-risk patients who are unlikely to deteriorate, thus enabling better ICU resource management and optimization.
The analytical software product uses AI-based algorithms and machine-learning models trained to identify the likelihood of occurrence of significant clinical events for patients in the intensive care unit (ICU). CLEWICU receives patient data from various sources, including Electronic Health Record (EHR) data and medical device data. The data is analyzed in near real-time to present calculated insights and notifications for dedicated AI models and provides a picture of overall unit status.
“We are proud to have received this landmark FDA clearance and deliver a first-of-its-kind product for the industry, giving healthcare providers the critical data that they need to prevent life-threatening situations,” said Gal Salomon, CLEW CEO.
“AI can be a powerful force for change in healthcare, enabling assessment of time-critical patient information and predictive warning of deterioration that could enable better informed clinical decisions and improved outcomes in the ICU,” said Dr. David Bates, Medical Director of Clinical and Quality Analysis in Information Systems at Mass General Brigham healthcare system and CLEW Advisory Board member.
“CLEW’s AI-based solution is a huge leap forward in ICU patient care, providing preemptive and potentially life-saving information that enables early intervention, reduces alarm fatigue and can potentially significantly improve clinical outcomes,” stated Professor Craig Lilly, University of Massachusetts Medical School.
About CLEW:
CLEW provides real-time AI analytics platforms designed to help providers make better informed clinical decisions by predicting life-threatening complications across various medical care settings. CLEW’s goal is to provide solutions that could improve outcomes and safety, streamline patient care, and efficiently handle regulations and penalties, ultimately lowering the cost of care. Originally developed and proven in the ICU, CLEW will develop machine learning models that have the potential to optimize clinical resources and guide health care providers in predicting patient deterioration, across all care settings. For more information, visit: www.clewmed.com
See Full Press Release at the Source: CLEW Medical Receives FDA Clearance for AI-Based Predictive Analytics Technology to Support Adult ICU Patient Assessment
Press Release by: CLEW Medical
 Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.


