Medical device and MedTech insights, news, tips and more
CoreLink Surpasses 5,000 Implants With Proprietary 3D Printed Technology
January 16, 2020
CoreLink, LLC, a leading designer and manufacturer of spinal implant systems, today announced the implantation of over 5,000 3D printed devices using their proprietary Mimetic Metal® technology.
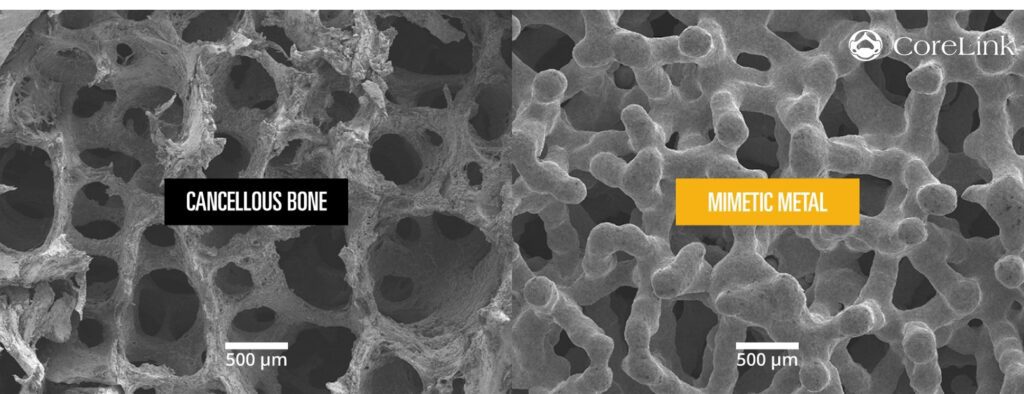
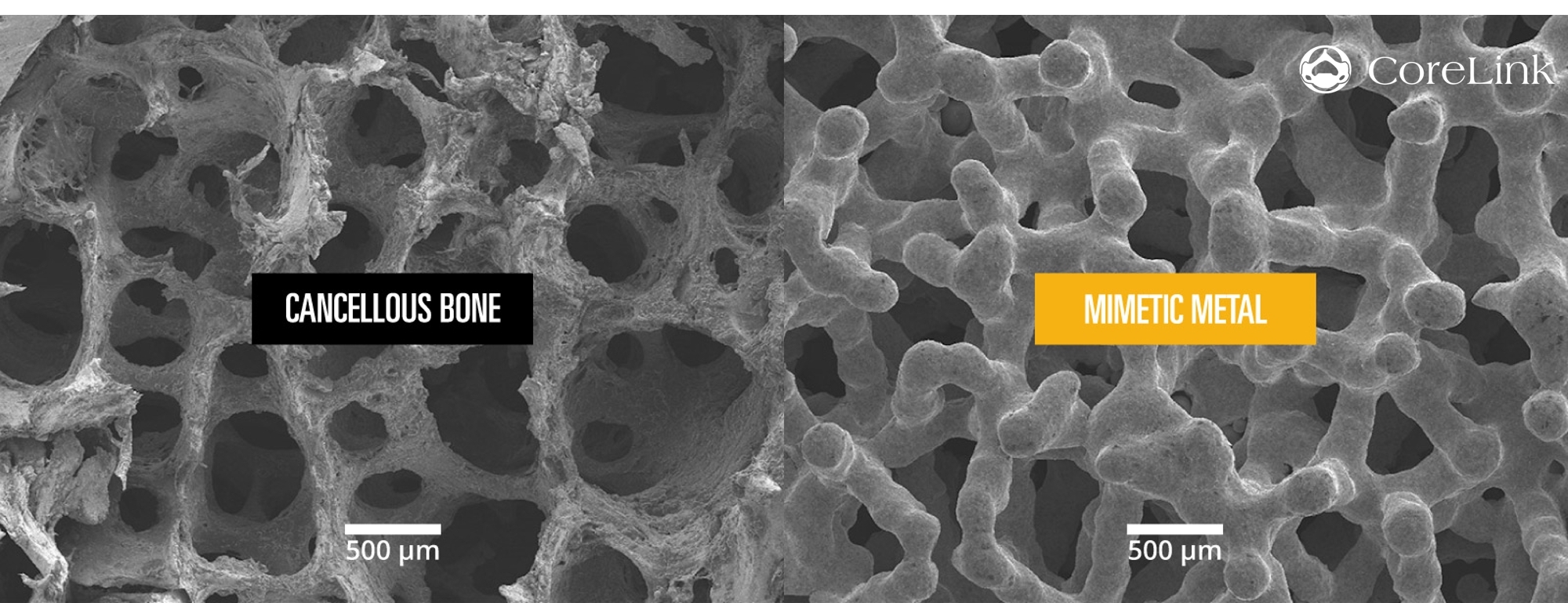
Mimetic Metal is an additively manufactured technology that combines a lattice framework and inner trabecular pores to emulate the structural, functional and physiological properties of bone. The unique dynamic design creates anisotropic stiffness to aid in load transfer. This encourages the structural adaptations of Wolff’s Law, which states that bone density will adapt to functional forces on the bone.
In addition, its open-pore architecture mimics cancellous trabeculae and provides hydrophilic wicking capabilities designed to allow blood flow through the implant. The streamlined titanium alloy (Ti-6AL-4V ELI) support structure also minimizes overall implant density, resulting in beneficial imaging characteristics.
“CoreLink set out to emulate nature with our 3D printed technology. Our unique design has enabled us to optimize everything needed in an interbody device – imaging, graft capacity, porosity, load sharing, strength, and stiffness,” said Jay Bartling, CEO, CoreLink. “From the development of the technology to execution of the 3D printing, we design and manufacture Mimetic Metal implants in our facility, ensuring both form and function are met to help patients in their healing process.”
CoreLink has a comprehensive offering of Mimetic Metal implants and exceptionally crafted instrumentation, including the CoreLink M3 Stand-Alone ALIF and F3D family of implants. F3D interbodies include curved, straight, anterior lumbar, cervical, and lateral implants in an array of footprints, heights, and lordosis.
CoreLink surpassed the 5,000th 3D printed device benchmark in November 2019. The first implantation was with the F3D anterior cervical interbody in June 2017.
About CoreLink
CoreLink, known as The Source for Spine™, internally designs and manufactures more than 99% of its broad portfolio of spinal implant systems. With a unique heritage that combines old-world craftsmanship with state-of-the-art manufacturing, we collaborate with surgeons to develop and deliver effective surgical solutions and improve the lives of patients. Learn more at corelinksurgical.com.
See Full Press Release: CoreLink Surpasses 5,000 Implants With Proprietary 3D Printed Technology | Business Wire
Written by: CoreLink, LLC

Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.