Medical device and MedTech insights, news, tips and more
Ezra Receives FDA Clearance for Prostate Cancer Artificial Intelligence
October 20, 2020
Ezra, the New York-based startup transforming early cancer screening using MRI, announced today that it has received FDA 510(k) clearance for its Artificial Intelligence designed to assist radiologists in analyzing and segmenting prostate MRI. Use of the innovative AI technology can help reduce the time and cost of MRI-based prostate cancer screening. It is the first Prostate AI to ever be cleared by the FDA.
As a result of Ezra implementing this new AI technology in its cancer screening service, Ezra is decreasing the price of the Ezra scan by 15%, to $575.
“Over the past two years, our team has worked tirelessly on building Ezra’s Prostate AI, and I’m thrilled to bring it to our imaging partners across the U.S.,” said Emi Gal, CEO and co-founder of Ezra. “We will continue to work towards making the interpretation of prostate MRI scans faster and more affordable, in order to support the millions of men who are at risk of prostate cancer.”
The Ezra AI achieves three important goals:
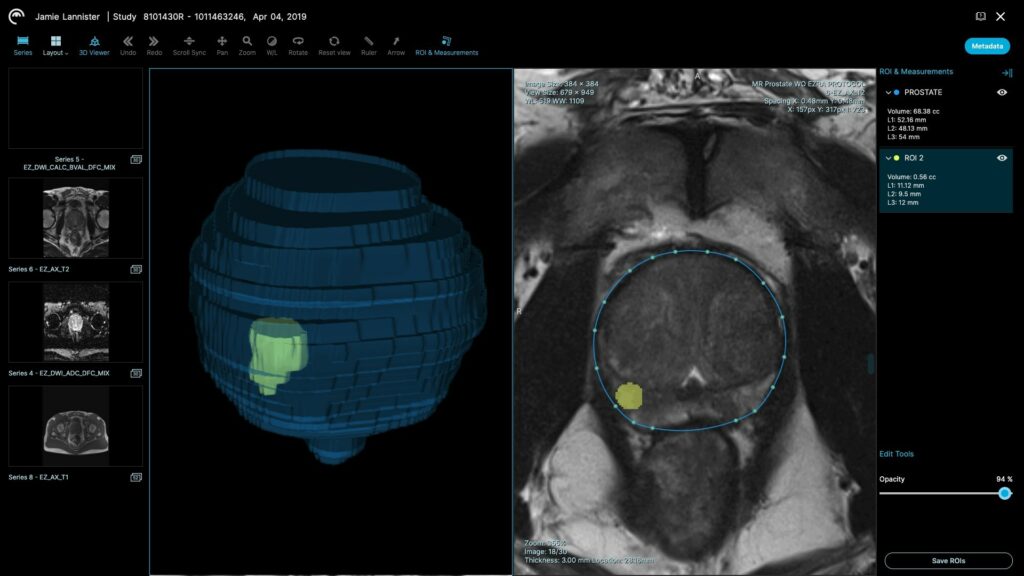
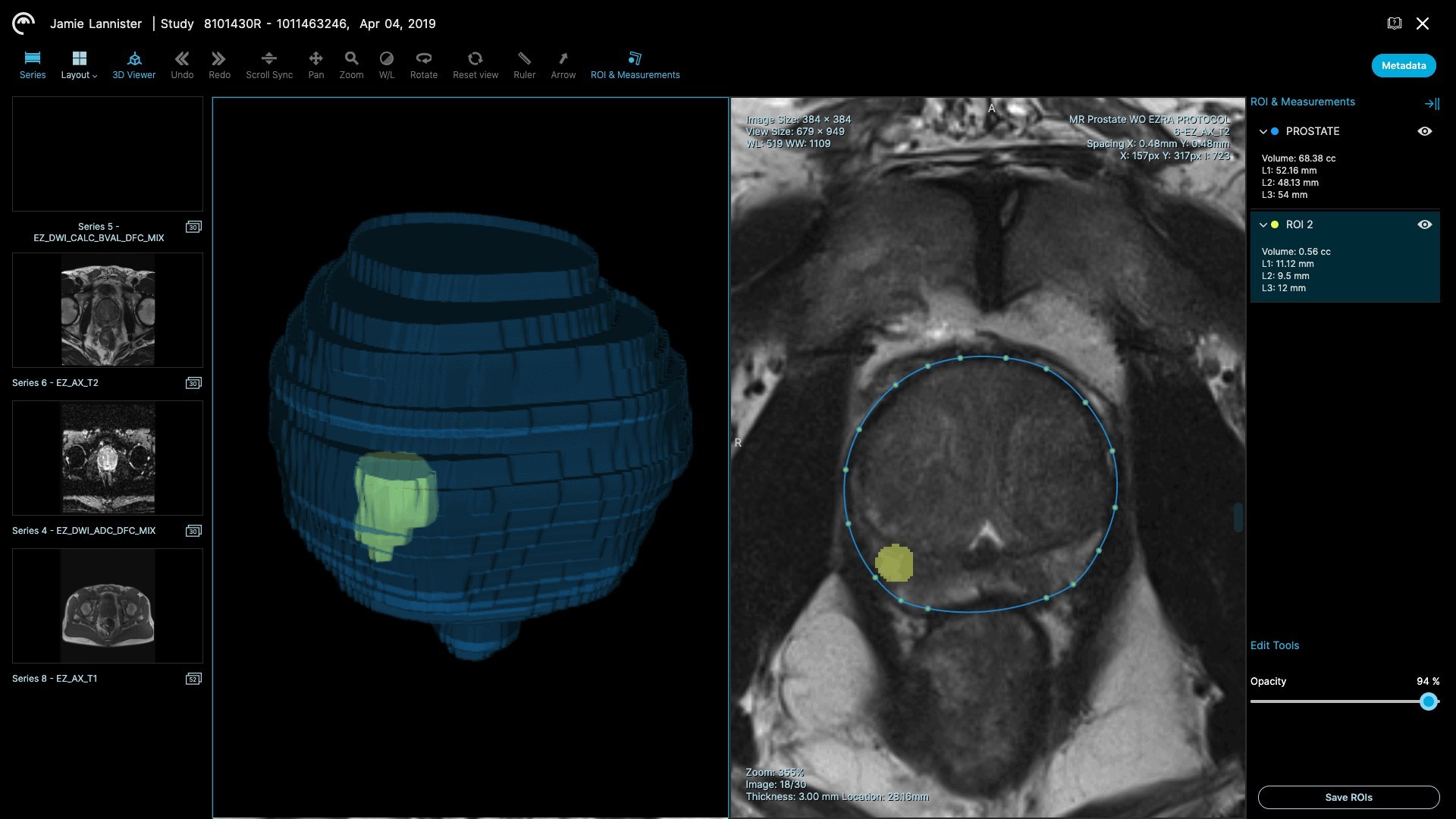
- Accurate prostate volume measurements. Accurate prostate volume is critical to screening because the size of the prostate is often an indication of potential disease.
- Automatic lesion quantification. Under current processes, if a lesion is identified in the prostate, a radiologist has to manually measure the size of the lesion, and grade it using the PI-RADS methodology. The Ezra Prostate AI helps radiologists segment lesions and can display the size of a lesion automatically, saving time, and minimizing patient worry.
- Automatic 3D volume rendering. Thanks to the Ezra Prostate AI’s segmentation capabilities, radiologists can automatically render 3D volumes of the prostate gland and lesions, which can improve their workflow efficiency.
To integrate the Ezra Prostate AI into radiology workflows, Ezra has also obtained FDA clearance for Plexo, a cloud-based PACS (Picture Archiving and Communications System) that works directly in the browser. This enables radiologists to use the Ezra platform without the need to install any software.
“Ezra is at the forefront of MRI-based cancer screening, and the company’s 510(k) FDA clearance for its Prostate AI is further validation of its innovation capabilities,” said Lawrence Tanenbaum, M.D., FACR, Vice president and CTO, Director of MRI, CT and Advanced Imaging at RadNet.
Ezra launched its MRI-based prostate cancer screening service in January 2019 and rolled out its full-body MRI scan in May 2019. Ezra partners with existing outpatient imaging facilities and all Ezra scans are analyzed by board-certified radiologists. The company’s cancer screening programs are live in New York City, San Francisco, and Los Angeles through its partnership with RadNet, Inc., the nation’s leader in outpatient imaging. In 2019, Ezra helped 4% of its members – all of whom were asymptomatic – detect cancer.
About Ezra
Ezra is a New York-based healthcare technology company utilizing medical imaging and Artificial Intelligence to provide individuals with full-body cancer screening that is fast, accurate, and affordable. By advocating for early cancer detection as part of annual health screenings, Ezra is on a mission to create a new standard of preventative care, providing people with the necessary knowledge to make decisions about their health. The company’s cancer screening services are available in New York, San Francisco, and Los Angeles.
See Full Press Release at the Source: Ezra Receives FDA Clearance for Prostate Cancer Artificial Intelligence