Medical device and MedTech insights, news, tips and more
FDA Clears Ninepoint’s OCT Imaging System for Use in Bile Ducts
August 14, 2019
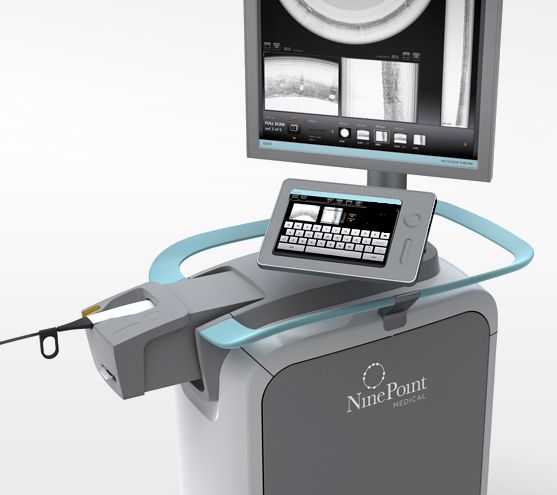
NinePoint Medical, Inc., a transformative medical device company pioneering the use of a real-time optical imaging platform for gastrointestinal applications, announced today that it has received U.S. Food and Drug Administration (FDA) clearance to market the NvisionVLE® Imaging System for use in the pancreas and bile duct. These anatomical indications add to the previously existing esophageal applications for NinePoint’s state-of-the-art Optical Coherence Tomography (OCT) imaging platform.
The NvisionVLE Imaging System provides physicians real-time, high-resolution volumetric images of the tissue surface and subsurface. This allows gastroenterologists to thoroughly evaluate tissue for regions of interest that may not be visible with other medical imaging technologies. The newly cleared application is associated with NinePoint’s Low-Profile Optical Probe, a 7 French diameter imaging probe designed to handle small and tortuous anatomies, such as those found in the pancreatico-biliary system. In the pancreas and bile duct, the ability to see beneath the tissue surface could help physicians answer a number of important clinical questions.
“When a patient enters our care with an undiagnosed condition in the pancreatico-biliary system, the stakes can be very high,” said Isaac Raijman, MD, previously the Chief of Gastroenterology at Baylor St. Luke’s Medical Center. “We have a number of tools today to help evaluate a patient’s condition within this anatomy. Unfortunately in many cases, our current toolset is unable to provide enough information to determine the best course of action. This is especially concerning for strictures or blockages of unknown origin. We are hopeful and excited that imaging beneath the surface with the NinePoint technology will supply the missing link and will enhance our clinical decision-making for these critically ill patients.”
This clearance marks another significant step for the strategic collaboration between NinePoint Medical and their exclusive worldwide distributor, Merit Medical Systems, Inc. “NinePoint continues to produce innovative and high quality products that our team has been very impressed with,” said Fred P. Lampropoulos, Chairman and CEO of Merit Medical. “The addition of new applications for the NvisionVLE Imaging System is important to demonstrate not just the quality but breadth of value the technology brings to hospitals and patients. We’re thrilled to add this to our suite of Merit Medical endoscopy products, and look forward to more great things from our collaboration with NinePoint.”
The pancreatico-biliary clearance marks a very productive 12 months of product development activity for NinePoint Medical. In November of 2018, NinePoint announced the first FDA-cleared artificial intelligence imaging technology for gastroenterology applications, the Intelligent Real-time Image Segmentation (IRIS) product. “I’m so proud of the team here at NinePoint,” commented Eman Namati, Ph.D., President and CEO of NinePoint Medical. “Our group over the years has really pushed the limits of engineering innovation to help patients. This past year has been especially fruitful, with the clearance and market launch of a first-of-its-kind artificial intelligence technology, and now with the clearance of a pancreatico-biliary application for the Low-Profile Optical Probe. The ability to promote to this new anatomical space really strengthens the platform of the NvisionVLE Imaging System, and continues to build the opportunity for this technology to help patients.”
See Full Press Release at the Source: NinePoint Medical Announces FDA Clearance for Pancreatic and Biliary Applications of the NvisionVLE® Imaging System | Business Wire
Press Release by Ninepoint Medical
 A Speciality Recruiting Firm Exclusively Servicing The Medical Device Industry
A Speciality Recruiting Firm Exclusively Servicing The Medical Device IndustryLegacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.

