Medical device and MedTech insights, news, tips and more
Make Way for Medrobotics: A Twist for Surgical Robotics
November 21, 2016

Since 2000, California-based Intuitive Surgical has dominated the world of robot-assisted surgery. Its da Vinci system has logged well over two million surgical procedures.
Now, however, a Massachusetts startup called Medrobotics has launched a system of its own that features a design that could make robots a much more common fixture in operating rooms.
Rather than been being limited to straight, “line-of-sight” procedures as in other surgical robots, the company’s Flex Robotic System enables surgeons, with full HD visualization, to guide the device through the twists and turns of the body’s pathways. Result: minimally invasive surgery can be performed on sites that were difficult or even impossible to reach before.

All the key components of the Flex Robotic System can be set up for surgery in about 10 minutes, say surgeons. The disposable Flex Drive (blue component pictured right) attaches to the Flex Base and is inserted into the patient.
“With this system, we can now reach areas, such as the top of the voice box, which were very hard to access,” says Eugene Myers, M.D., a head and neck surgeon and professor at the University of Pittsburgh Medical School. “For example, this can mean the difference between a person losing his voice or retaining it.”
So far, the Flex Robotic System has received both U.S. Food and Drug Administration approval and Europe’s CE mark for transoral procedures of the mouth and throat. In October 2016, the system also gained European clearance for colorectal surgeries, expanding the range of operations. Costing about $1 million–around half the price of the latest da Vinci model–the system offers a relatively small footprint and a reported 10-minute setup time in operating rooms.
The device is gaining recognition, including a Best of Show award in the 2016 Medical Design Excellence Awards, and a 2016 Red Herring Top 100 Award, which recognizes technologies most likely to change people’s lives. Meanwhile, the technology has attracted about $150 million in investment for Medrobotics.
“The global market for robotics and other tools for minimally invasive surgery is expected to double by 2020,” says James Jordan, chief investment officer for Pittsburgh Life Sciences Greenhouse, an early investor in Medrobotics. “But that type of increase is not likely to occur without flexible robotics technology.”
Roots at Carnegie Mellon
The Medrobotics story began at Carnegie Mellon University’s Robotics Institute, which has spawned robotics innovations ranging from devices to inspect power plants and explore space environments to autonomous tractors and self-driving cars. (Read Part 1 and Part 2 of our conversation with the Institute’s legendary William “Red” Whittaker.)
 In the late 1990s, Robotics Institute engineering professor Howie Choset (pcitured right) became fascinated with what has become known as “snake robots,” highly-articulated mechanisms with many degrees of freedom. These robotic snakes can thread their way through tightly packed areas in such applications as search and rescue missions and the manufacture of giant aircraft wings.
In the late 1990s, Robotics Institute engineering professor Howie Choset (pcitured right) became fascinated with what has become known as “snake robots,” highly-articulated mechanisms with many degrees of freedom. These robotic snakes can thread their way through tightly packed areas in such applications as search and rescue missions and the manufacture of giant aircraft wings.
Along with fellow CMU engineering professor Alon Wolf and heart surgeon Marco Zenati, M.D., Choset developed the first prototype of a snake robot for minimally invasive surgery.
Together, the three men founded Medrobotics in 2005. They initially targeted the device for heart surgery, and it was used successfully in a limited investigational trial on three patients in Europe in 2010. It became apparent, however, that the high cost of developing a commercial device for cardiac applications, as well as the regulatory hurdles, dictated moving to other surgical procedures for faster commercial launch.
Noting that commercializing a surgical device is “not something that a robotics professor can accomplish with grants to the university,” Choset knew it was time to hand off his invention to Medrobotics. The company not only had to raise an enormous amount of capital for development and manufacturing infrastructure, but its engineers had to refine Choset’s design, both to pass muster with regulators and to meet the clinical demands of surgeons.
“The genius of Professor Choset’s design was his revolutionary motion system, specifically the relationship of the inner and outer mechanisms of the robot that allows the articulation to occur, just like a snake,” says Samuel Straface, a Ph.D. and former scientist who serves as president and CEO of Medrobotics. “Our engineers had to transfer that know-how into a viable product for clinicians.”
Follow the Leader
The Flex Robotics System that received FDA clearance in July 2015 combines the benefits of a laparoscope – a rigid, straight-viewing device used in minimally-invasive surgery — with an endoscope, a flexible instrument for visual diagnosis.
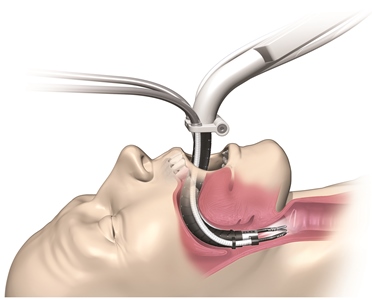 This illustration shows the key advantage of the Flex Robotic System: the ability to navigate through the body’s twists and turns to hard-to-reach surgical sites.
This illustration shows the key advantage of the Flex Robotic System: the ability to navigate through the body’s twists and turns to hard-to-reach surgical sites.
Much of the device’s intellectual property revolves around the disposable Flex Drive, which snaps into a reusable base that contains system motors and controls. For transoral applications, the portion of the 1.5-lb drive inserted into the patient and containing the camera measures about 17 cm long, with a diameter that ranges from 17.5 mm to a maximum of 28 mm at the “distal end” where the surgery takes place.
The drive contains an external cable for electrical connections to the distally mounted, detachable camera and LEDs, plus an internal lumen that serves as a channel for tubing that carries fluid to a lens washer for the camera. The Flex Drive also contains accessory channels on either side of the camera to accommodate miniature instruments that the surgeon inserts during an operation.
At the heart of the Flex Drive’s technology is its motion system, which features a series of articulated segments or links. The design includes both inner and outer articulated segments, made of medical-grade polymer and arranged in a concentric mechanical assembly.
See Video: TVEyes Media Monitoring Suite – [Transcript]
Read Full Article – Source: A Twist for Surgical Robotics | Engineering360
Author – Larry Maloney
