Medical device and MedTech insights, news, tips and more
Med-botics Wearable Device Detects Opioid Overdose, and can Provide Early Warnings to Over-sedated Patients
November 6, 2019
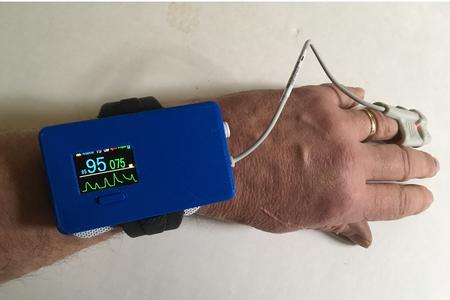
The FDA granted Breakthrough Device status to Med-botics, LLC, (www.med-botics.com), for its Oxalert EPOTM (Enhanced Pulse Oximeter) device. The wrist-worn arousal device is designed to prevent respiratory arrest and death from opioid overdose. FDA Guidance documents describe Breakthrough Devices as “innovative technologies that diagnose or treat life-threatening conditions more effectively than any FDA-approved products.”
The FDA says the Oxalert is “intended to continuously monitor SpO2 (blood oxygen levels) in post-operative in-patients and home-users receiving opioids and to arouse them to breathe if SpO2 decreases below 90%.”
A 2011 proof-of-concept study done at Oregon Health Science University (OHSU) showed that a continuous pulse oximeter, headphones for verbal prompting, a nerve stimulator, and a laptop computer automatically prevented hypoxemia (oxygen levels below 90%) in heavily-sedated patients emerging from surgery.
Med-botics, a physician-owned start-up, licensed the patented method from OHSU and re-invented the system that actually needed to be wheeled on a cart. The Oxalert EPO arousal device exists now as a simple user-friendly wrist-worn battery-operated arousal system.
Only about 10% of the 40 million post-op in-patients receiving opioids in the U.S. are continuously monitored. A 2015 study found that hypoxemia is commonly unrecognized in post-operative in-patients without continuous monitoring and incidentally, that hypoxemia normalizes with arousal (the Oxalert device method).
Nearly all of the 80,000 deaths yearly from opioid overdose occur at home. Opioids use after surgery usually continues at home after hospital discharge. Med-botics aims to make opioids safer both in-hospital and at home.
Dr. Richard Dutton, former Executive Director of the Anesthesia Quality Institute, says, “Providing early warning to over-sedated patients and their caregivers is important in the hospital and even more important at home. I believe this is a device that will save lives. I am looking forward to commercial availability of this product as an important step in the effort to eradicate ‘dead-in-bed’ adverse events, as well as the many other sequelae of opioid-induced post-operative hypoxemia.”
See Full Press Release: Oxalert EPO™ Wearable Device Granted FDA Breakthrough Status
Written by: Med-Botics

Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.
