Medical device and MedTech insights, news, tips and more
Non-Surgical Weight Loss Balloon System: Interview with Obalon Therapeutics CEO
December 5, 2016
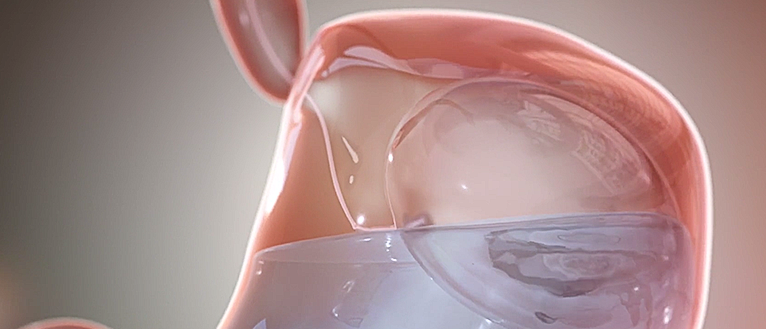
Medgadget recently reported on the FDA approval of the Obalon balloon system, which assists weight loss in obese patients who have failed to lose weight by other means and is an alternative to conventional gastric bypass surgery. The system has been developed by Obalon Therapeutics, a company out of Carlsbad, California, and consists of inflatable balloons that can be inserted into the stomach in a minimally invasive manner. The treatment involves swallowing a deflated balloon in the form of a small capsule attached to a thin tube. Once in the stomach, the balloon is inflated and the tube is withdrawn. A maximum of up to three balloons can be placed in the stomach, to be left in place for up to six months before deflation and removal using an endoscopic procedure. The balloons contribute a feeling of satiety, meaning that patients tend to eat less and lose weight.
Medgadget had the opportunity to ask Andy Rasdal, CEO of Obalon Therapeutics some questions about the Obalon balloon system.
Conn Hastings, Medgadget: Congratulations on the recent FDA approval of the Obalon system. Can you give us a brief overview as to how the system compares with other treatments for obese patients?
 Andy Rasdal, Obalon Therapeutics: Thank you. The FDA PMA approval of the Obalon Balloon System came only after rigorous FDA review and the successful completion of a double-blinded, randomized, sham-controlled trial that met its co-primary weight loss endpoints and demonstrated a strong safety profile. The Obalon Balloon System (the “System”) is a swallowable intragastric balloon system indicated for temporary use to facilitate weight loss in adults with obesity (BMI of 30 – 40 kg/m2) who have failed to lose weight through diet and exercise.
Andy Rasdal, Obalon Therapeutics: Thank you. The FDA PMA approval of the Obalon Balloon System came only after rigorous FDA review and the successful completion of a double-blinded, randomized, sham-controlled trial that met its co-primary weight loss endpoints and demonstrated a strong safety profile. The Obalon Balloon System (the “System”) is a swallowable intragastric balloon system indicated for temporary use to facilitate weight loss in adults with obesity (BMI of 30 – 40 kg/m2) who have failed to lose weight through diet and exercise.
The Obalon Balloon is a very different technology platform than the other two gastric balloons currently approved in the U.S. – the ReShape Duo and the Apollo Orbera. For ReShape and Apollo, a single balloon is placed in the stomach via an endoscopic procedure under anesthesia and filled with liquid. In contra st, the Obalon Balloon is swallowed in a dissolvable capsule without the need for anesthesia and filled with gas. Up to three balloons may be swallowed and placed during the therapy period. At Obalon, our design intent was to create a gastric balloon that minimized side effects and provided progressive weight loss over the full six-month treatment period.
Medgadget: Gastric bypass surgery produces a permanent change in the architecture of the gastrointestinal tract, whereas the balloon system is removed after 6 months. Is there a risk that patients could regain weight once the system is removed, or do lasting changes in lifestyle occur?
Andy Rasdal: We studied that in our U.S. Pivotal Trial that support FDA approval and the data was presented on November 4, 2014 by Dr. Aurora Pryor in a scientific session at Obesity Week. Dr. Pryor presented that at the end of one year, six months after the balloons were removed, patients had maintained 89.5% of their weight loss on average.
Medgadget: How was the balloon system conceived?
Andy Rasdal: We started Obalon more than eight years ago with the intent to invent a novel, non-surgical and fully reversible device that would give overweight and obese people a safe and effective option to reverse the progress of this chronic disease. Obalon did not invent the idea of a space-occupying device for weight loss, or even concept the gastric balloon, but Obalon has invented the first and only swallowable, gas-filled balloon approved by the FDA.
The Obalon Balloon is intended to occupy space in the stomach to make patients feel full sooner, eat less and lose weight. In addition to the strong safety and efficacy demonstrated in our US Pivotal Trial that supported our FDA approval, we believe the Obalon Balloon offers many important ease-of-use advantages over prior and current gastric balloons to both providers and patients.
First, by being swallowable, the Obalon Balloon is easy to place – a patient simply swallows the capsule with the balloon inside without anesthesia or sedation. Placement does not require an endoscopic procedure under anesthesia like other gastric balloons. The simple placement enables balloon volume to be increased incrementally over the treatment period by simply swallowing up to two more additional balloon capsules, which is intended to minimize side effects and create progressive weight loss over the full treatment period.
Next, by being gas-filled, the balloon is light and floats up in the fundus, or top of the stomach, as opposed to sinking to the antrum, or bottom of the stomach, like other prior and current gastric balloons. This is intended to be less offensive to the stomach to increase patient comfort and reduce the adverse events seen with conventional liquid-filled balloons.
Creating a swallowable, gas-filled balloon required the invention of first of its kind technology. Our multi-layer balloon film is thin enough to fit into a swallowable capsule, but robust enough to withstand the chemical and mechanical forces of the stomach trying to digest it. Our balloon is filled with a proprietary mix of gas, which along with a novel inflation valve, ensure full balloon inflation throughout the full six-month treatment period. Finally, we have developed unique capsule and catheter technology that enables the balloon to be reliably delivered to and inflated in the stomach.
Read Full Article – Source: Obalon Non-Surgical Weight Loss System: Interview with Andy Rasdal, CEO of Obalon Therapeutics. |
Author – CONN HASTINGS
