Medical device and MedTech insights, news, tips and more
PatenSee’s Contactless Imaging Device Shows Promise in Monitoring Dialysis Patients
February 24, 2022
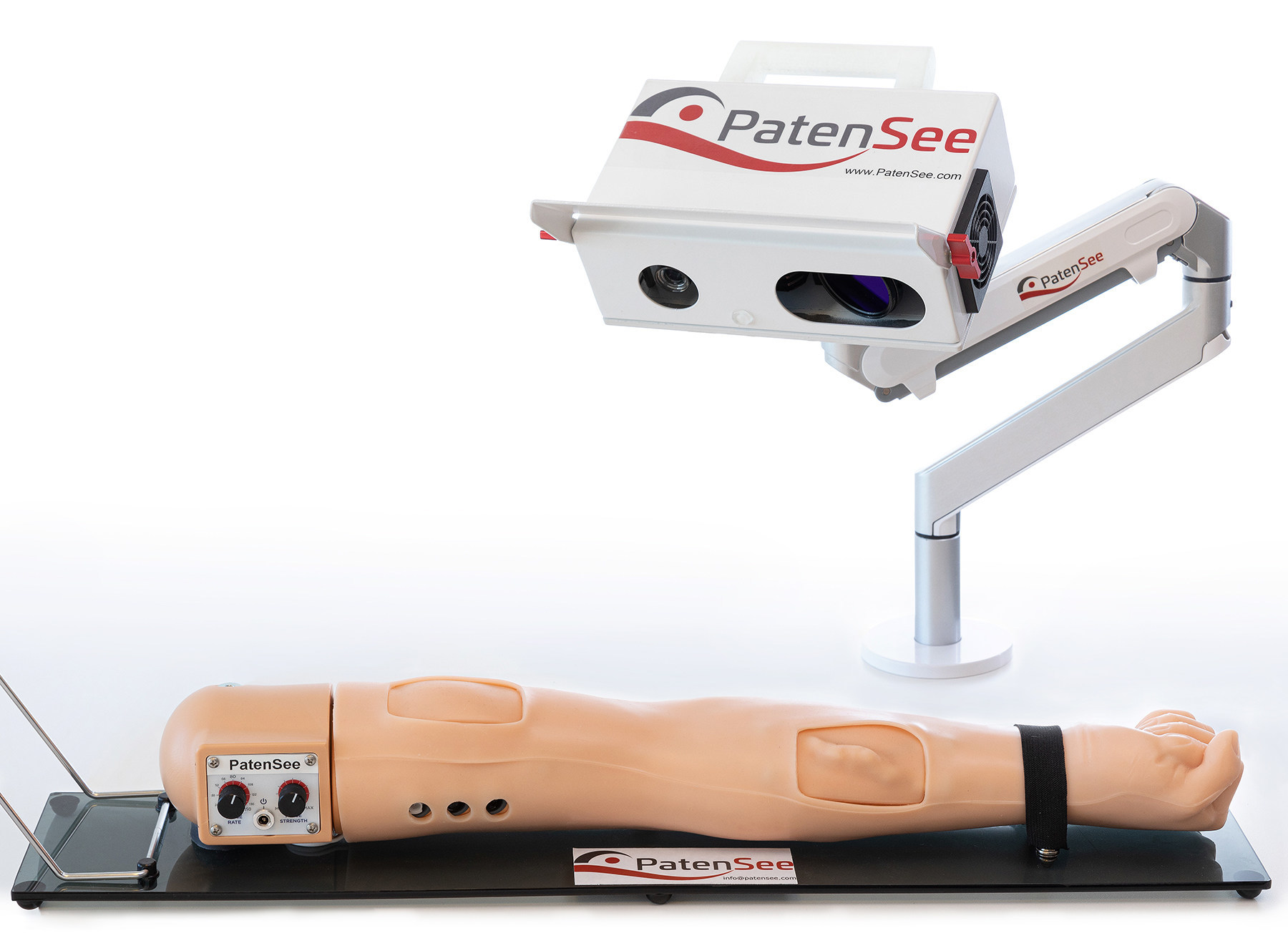
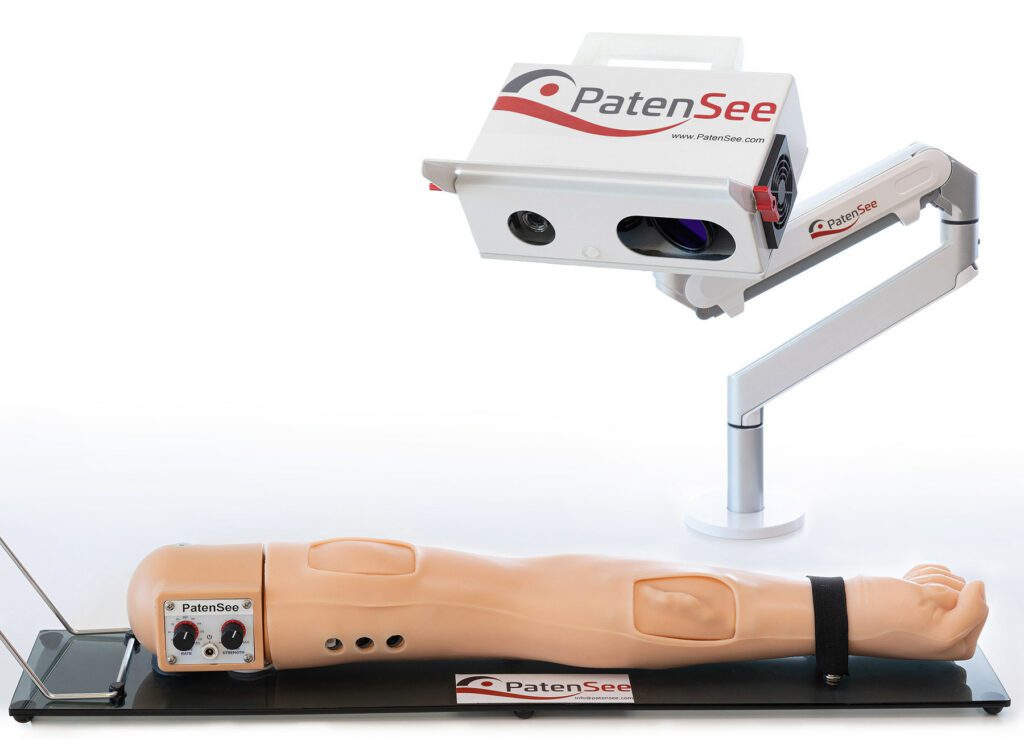
The interim results of an ongoing clinical study of PatenSee‘s contactless imaging device for the early detection of vascular access stenosis for dialysis patients were shared in a poster presentation. The presentation, titled First Clinical Experience with Non-Invasive, Contactless, Optical Surveillance of Vascular Access, was delivered by the study’s principal investigator, Professor Benaya Rozen-Zvi, MD at the 2022 World Congress of Nephrology Meeting in Indonesia.
The vascular access fistula (AVF) connects the patient, usually from their arm, to the dialysis machine. This makes it critically important to protect the AVF from clogging. A blocked access can cause various complications including loss of the AVF and death. The goal is to detect initial clogging before it leads to an unwanted patient outcome, but ongoing physical monitoring by the medical team is a major challenge. PatentSee’s imaging technology is designed to perform quick, high-quality monitoring without any physical contact with the patient, thus reducing risk of infection, especially important in pandemic times.
“Avoiding AVF stenosis has never been more important. Dialysis treatment centers are very busy, physical examinations take time and require touching an already sensitive area on the patient, plus there is the risk of infection from contact, especially in our new COVID reality. The development of a contactless system that can nearly instantly provide accurate information of an AVF’s status couldn’t have come sooner and the clinical results thus far demonstrate that PatenSee’s technology has strong potential to ease the burden of AVF monitoring,” said PatenSee CEO Dr. Gal Goshen.
Thirty-six dialysis patients participated in more than 600 sessions collectively at Rabin Medical Center in Israel to test the efficacy and timeliness of PatenSee’s imaging device.
Imaging took approximately two minutes and was performed during the waiting time prior to the dialysis sessions. A comparison to a doppler ultrasound done by a technician blinded to the results of PatenSee’s device demonstrated good correlation suggesting that this rapid, contactless, machine learning, imaging device could serve as an effective tool for automated AVF surveillance.
About PatenSee
PatenSee, a portfolio company of the MEDX Xelerator, is a clinical-stage medical device company developing a contact-free imaging surveillance system for the early detection of vascular stenosis in hemodialysis patients. Using advanced, multi-modal imaging technologies and machine learning, PatenSee’s device aims to alert regarding stenosis risk at the earliest stage, enabling timely interventions to protect the patient’s hemodialysis lifeline, improve quality of care and prolong life expectancy in both the clinic and the home dialysis setting. The dialysis market is expected to exceed $100B by 2024. PatenSee is currently raising a series A to fund the next stages of its clinical program towards the commercial launch of the product. For more information, please visit www.patensee.com.
See Full Press Release at the Source: PatenSee’s Contactless Imaging Device Shows Promise in Timely and Accurately Monitoring Dialysis Patients
Press Release by: PatenSee
Legacy MedSearch has more than 30 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 10 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.