Medical device and MedTech insights, news, tips and more
Stimdia Medical Inc. Initiates FDA Approved IDE Pivotal Trial to Investigate Impact of Neuromuscular Stimulation on Mechanically Ventilated Patients
October 4, 2023
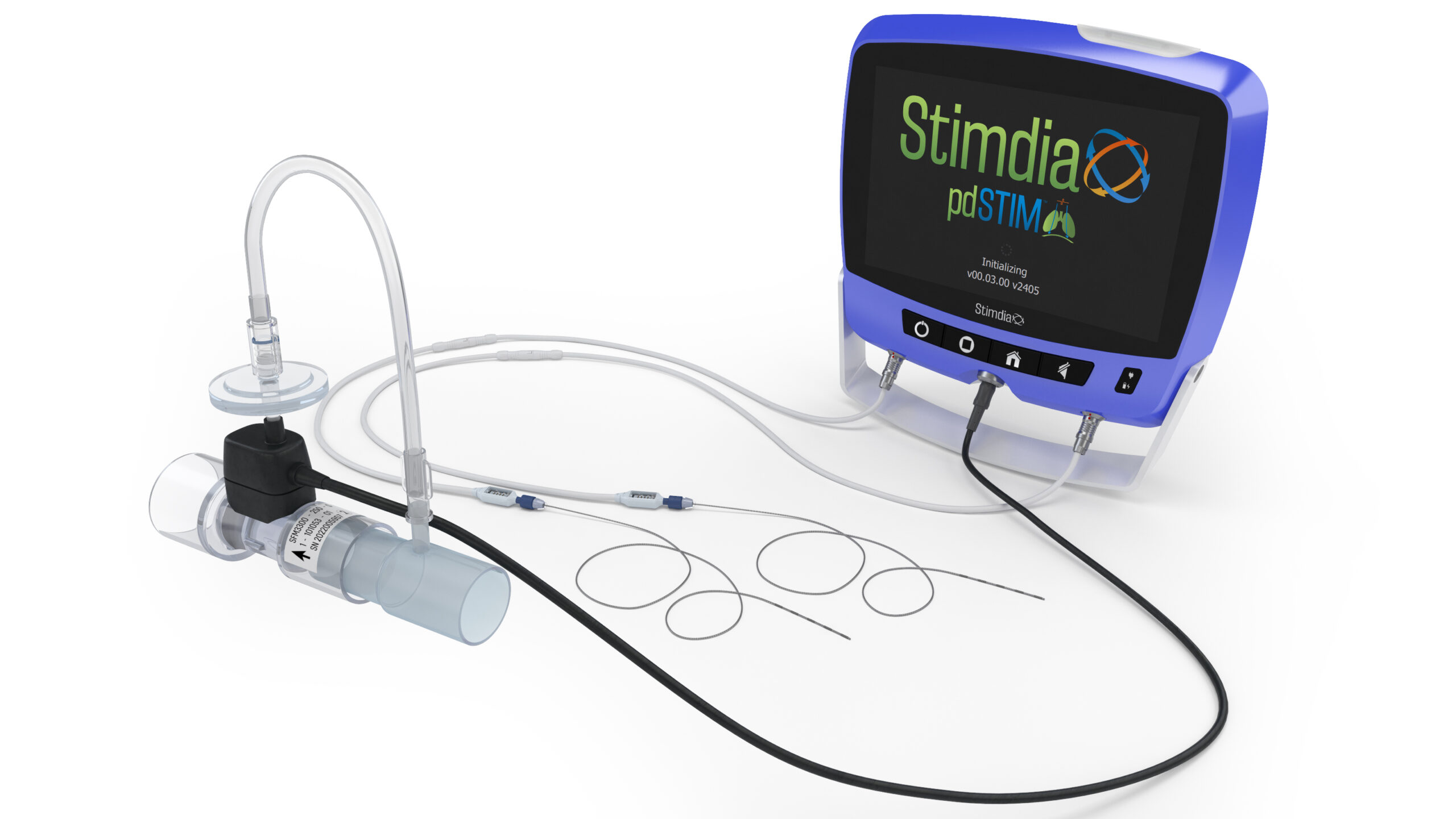
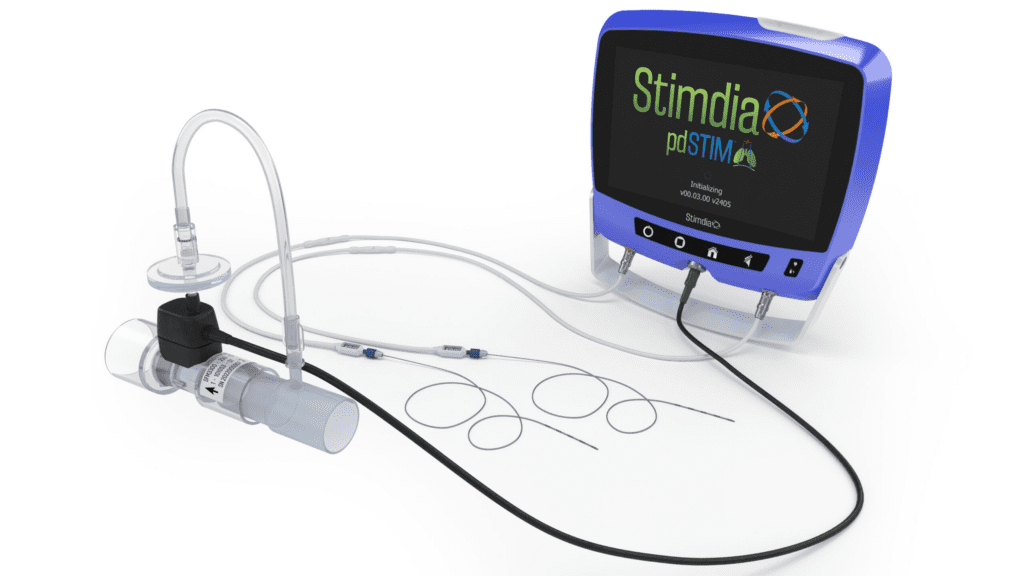
Stimdia Medical, a medical device company developing innovative technologies to improve respiratory care, today announced the initiation of its FDA approved pivotal trial to investigate the impact of phrenic nerve stimulation (PNS) on the time to liberate patients from mechanical ventilation. The ReInvigorate Study is a randomized, controlled trial that will enroll approximately 420 patients who are receiving mechanical ventilation in the intensive care unit (ICU). Patients will be randomly assigned to receive PNS therapy utilizing Stimdia’s proprietary pdSTIMTM System or standard of care alone. The primary efficacy endpoint of the ReInvigorate Study will evaluate the time to liberation from mechanical ventilation.
Dr. Kyle Cunningham, trauma and critical care physician at Atrium Health – Carolinas Medical Center had this to say about the therapy: “We are excited to have enrolled the first subject in the ReInvigorate Study. It is great to be able to investigate this new therapy in our patients who are having difficulties regaining independent breathing. This intuitive, minimally invasive approach has the potential to reverse some of the modalities that are all too common with existing ventilation techniques. We are hopeful that this therapy will make a significant difference in the lives of our patients.”
Dr. Steven Conrad, Louisiana State University, Division of Critical Care Medicine, the National Principal Investigator for the ReInvigorate Study, commented, “The pdSTIM System has the potential to introduce a novel approach to ICU patient management, assisting physicians, respiratory therapists, and nurses in expediting the liberation of patients from mechanical ventilation. I look forward to clinicians engaging in this therapy as part of the study and await the trial’s outcomes as it progresses toward completion.”
The pdSTIM System uses a minimally invasive procedure to place small leads adjacent to the phrenic nerves on each side of the neck. These leads are connected to an external console that synchronizes the delivery of therapy with inhalation. The device can be programmed to deliver electrical impulses at a specific frequency and intensity. These electrical impulses are intended to awaken and recondition the diaphragm, the primary muscle used to support breathing. Strengthening the diaphragm is believed to be an avenue towards weaning patients from mechanical ventilation more quickly.
“We are thrilled to partner with an outstanding group of clinicians to launch this pivotal trial that will investigate the potential of PNS to improve the outcomes of critically ill patients,” said Stimdia Medical CEO Tim Miller. “We believe that the pdSTIM therapy has the potential to revolutionize respiratory care, and we are dedicated to making this therapy accessible to patients as soon as possible.”
The pdSTIM System is an investigational device and is limited by federal (or United States) law to investigational use and is not available for sale or commercial use.
About Stimdia Medical
Headquartered in Minnesota, Stimdia Medical, has developed the pdSTIM System to facilitate weaning patients from mechanical ventilation through the use of neuromuscular stimulation to the phrenic nerves. The device is currently for investigational use only and is not for sale in any country.
For more information about the ReInvigorate Study, please visit the Stimdia Medical website at www.stimdia.com or at Clinicaltrials.gov (NCT05998018).
See Full Press Release at the Source: Stimdia Medical Inc. Initiates FDA Approved IDE Pivotal Trial to Investigate Impact of Neuromuscular Stimulation on Mechanically Ventilated Patients
Press Release by: Stimdia Medical
Legacy MedSearch has more than 35 years of combined experience recruiting in the medical device industry. We pride ourselves on our professionalism and ability to communicate quickly and honestly with all parties in the hiring process. Our clients include both blue-chip companies and innovative startups within the MedTech space. Over the past 17 years, we have built one of the strongest networks of device professionals ranging from sales, marketing, research & , quality & regulatory, project management, field service, and clinical affairs.
We offer a variety of different solutions for hiring managers depending on the scope and scale of each individual search. We craft a personalized solution for each client and position with a focus on attracting the best possible talent in the shortest possible time frame.
Are you hiring?
Contact us to discuss partnering with Legacy MedSearch on your position.