Archive for August 2016
Spinal Simplicity Introduces Next Gen Spine Fusion Device With New Feature
LEAWOOD, Kan., Aug. 15, 2016 /PRNewswire/ — Spinal Simplicity, a medical device company based in Overland Park, Kan., that develops innovative solutions to treat complex surgical problems, recently announced a new generation of their Minuteman G3 fusion device that features an FDA-cleared coating of hydroxyapatite (HA). “HA has been used in orthopedic procedures for the last…
Read MoreListings for Heart Transplants Increase Since ACA Implementation
Since the implantation of the Patient Protection and Affordable Care Act (ACA) in 2014, listings for heart transplants have increased by 17 percent, according to a recent analysis. Meanwhile, listings for liver transplants and kidney transplants only increased 2 percent and 1 percent, respectively, during the same time period. Lead researcher Guilherme H. Oliveira, MD,…
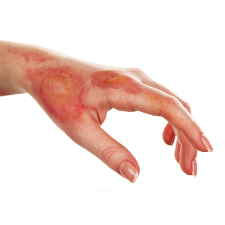
Read MoreNovel Technology May Prevent Burn Scarring
A group of researchers from Tel Aviv University and Harvard University has devised a new non-invasive method to prevent burn scarring caused by the proliferation of collagen cells. They are using short, pulsed electric fields prevent the formation of burn-related hypertrophic scars — raised tissue caused by excessive amounts of collagen. Research for the study…
Read MoreJury Sides with Surgeon in Bone Cement Trial
SEATTLE (AP) — A jury has found in favor of a University of Washington surgeon who was sued by the daughter of a patient who died after the doctor used a non-FDA-approved bone cement during the woman’s spinal surgery, a lawyer said. In a 10-2 decision after a five-week trial, the jurors reached the decision…
Read More5 Communication Skills Every Engineer Needs
There was a time when engineers could work within companies and have minimal (or even bad) communication skills. While it is not necessarily important for engineers to become great public speakers or authors, it is increasingly important for them to possess effective communication skills. Here’s why. Engineers Need to Sell Engineers do not necessarily need…
Read MoreA deep flaw has been discovered in thousands of neuroscience studies using fMRI
Modern neuroscience would be impossible without functional magnetic resonance imaging, or fMRI. The technique is barely 25 years old, but thousands of studies that use it are published each year. When you see headlines such as “Vegetative state patients can respond to questions” or “This is your brain on writing,” you can be sure that…
Read MoreFDA issues new draft recommendations for medical device manufacturers
The FDA has published draft updated recommendations that assist manufacturers with knowing when they are legally required to notify the FDA about medical device modifications. An additional guidance focused on software devices was also published. The two documents, once finalized, will replace a guidance that dates back to 1997. “These draft recommendations are intended to…
Read MoreMedicrea Announces €20 million Financing to Support Commercial Expansion of UNiD™ Spine Rods
Medicrea Appoints Richard Kienzle, Founding Member of Globus Medical, as Board Director, Chief Commercial Officer and Business Development Officer, while Athyrium Capital Management Purchases €15 million in Convertible Bonds with Additional €5 million Raised Through Equity Private Placement for the spread of Medicrea UNiD rods. NEW YORK, NY and LYON, France (August 11, 2016) –…
Read MoreWhy Have A Dog-Friendly Office? See Ours Plus the Country’s Top 11
Our office mascot, Jackson, easily makes Legacy MedSearch a top dog-friendly office! It’s more than a trend: it’s science. Spending time with dogs can increase productivity, workplace happiness, even prolong your life—and offices from coast to coast are taking note. After all, who doesn’t want a happier, healthier workforce (and lots of puppy kisses, to boot)?…
Read MoreHealthcare Fraud: Johnson & Johnson Subsidiary Pays $18 Million in Settlement
BOSTON, MA–A Johnson & Johnson subsidiary was involved with skirting U.S. Food and Drug Administration (FDA) rules for its own gain, allegedly placing profit ahead of patient safety. Acclarent Inc., J&J’s California-based manufacturer of medical devices, paid $18 million to resolve federal allegations that it caused health care providers to submit false claims to Medicare and…
Read More