Archive for August 2020
Synchron’s Stentrode Brain-Computer Interface Receives Breakthrough Device Designation from FDA
Synchron, a neurovascular bioelectronics medicine company, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device designation for Stentrode, a fully-implantable medical device that can translate brain activity or stimulate the nervous system from the inside of a blood vessel, without the need for open brain surgery. The device, which has…
Read MorePhilips to Acquire Intact Vascular for $275M
Royal Philips, a global leader in health technology, today announced that it has signed an agreement to acquire Intact Vascular, Inc., a U.S.-based developer of medical devices for minimally-invasive peripheral vascular procedures. Intact Vascular will enhance Philips’ image-guided therapy portfolio, combining Philips’ interventional imaging platform and diagnostic and therapeutic devices with Intact Vascular’s unique, specialized…
Read MoreFDA Clears SurGenTec Synthetic Bone Graft
SurGenTec, a privately held spine and orthopedic technology company, announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its proprietary OsteoFlo NanoPutty- Quadphasic Synthetic Bone Graft. OsteoFlo NanoPutty is a novel bone graft solution featuring the world’s first, and only quadphasic synthetic bone graft particles with nano-surface…
Read MoreHologic Buys Accessa Health for $80M
Hologic, Inc., a global leader in women’s health, announced today that it has acquired Acessa Health Inc., a privately-held innovator in minimally invasive treatment for fibroids, for approximately $80 million in cash plus contingent payments based on future revenue growth. “Acquiring Acessa Health strengthens our leadership position in the GYN surgical space and broadens our…
Read MorePelvital Announces FDA Clearance of Flyte Pelvic Floor Device
Pelvital, a medical technology company focused on women’s health, announced it has received U.S. Food and Drug Administration (FDA) clearance for Flyte, a first-of-its-kind, non-invasive, intravaginal home-use device that is intended for strengthening of the pelvic floor muscles, which has been found to help women with stress urinary incontinence. According to the National Association for…
Read MoreValencia Technologies Files Pre-Market Approval Application with U.S. FDA for eCoin Peripheral Neurostimulator
Valencia Technologies Corporation (“Valencia“), a private medical device company, today announced the filing of its pre-market approval (PMA) application of its eCoin® Peripheral Neurostimulator System for overactive bladder with the Food and Drug Administration (FDA). eCoin® is a first-in-kind neurostimulator providing intermittent electrical stimulation of the tibial nerve via a leadless nickel-sized and shaped device fully-implanted under…
Read MoreHyperfine Receives FDA 510(k) Clearance for Portable MRI System
Hyperfine Research, Inc. has received 510(k) clearance from the US FDA for its category-defining portable MRI technology, the Swoop™ Portable MR imaging device. Hyperfine’s Swoop™ system is a point-of-care MR imaging device that wheels directly to the patient’s bedside, plugs into a standard electrical wall outlet and is controlled through a wireless tablet, making MR imaging…
Read MoreCeek Women’s Health Introduces Nella NuSpec, a Smarter Reusable Vaginal Speculum
Ceek Women’s Health, a groundbreaking women-led medical device company, is excited to announce its latest innovation, the Nella NuSpec Reusable Vaginal Speculum. The Nella NuSpec was developed in partnership with OBGYN providers and patients through an extensive 5-year R&D process. The reusable vaginal speculum commonly used today is made from metal and has not changed…
Read MoreMagVenture Receives FDA Clearance for OCD
FDA has cleared MagVenture TMS Therapy for adjunct treatment of Obsessive-Compulsive Disorder (OCD). This marks the second indication in the US for the Danish medical device company MagVenture who is already FDA cleared for the treatment of major depressive disorder. OCD is a mental health disorder characterized by unreasonable thoughts and fears (obsessions) which lead…
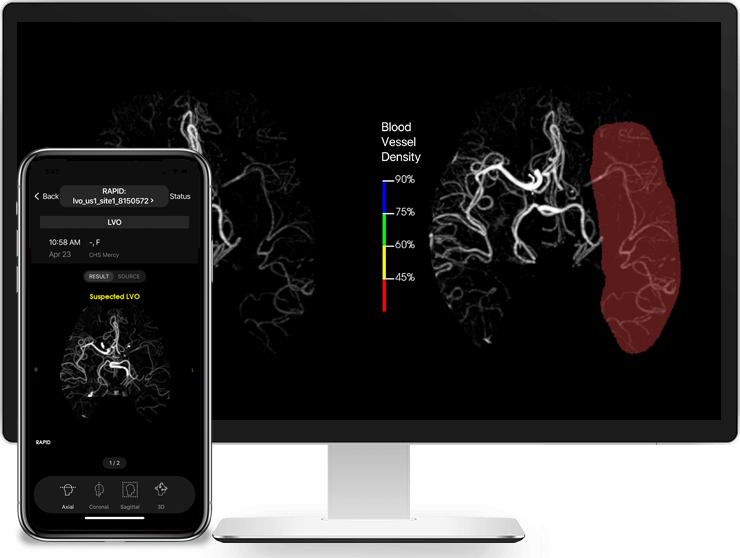
Read MoreRapidAI Receives FDA Clearance of Rapid LVO For Identification of Suspected Large Vessel Occlusions
RapidAI, the worldwide leader in advanced imaging for stroke, today announced that Rapid LVO has received Food and Drug Administration (FDA) clearance for detecting suspected LVOs (Large Vessel Occlusions). Rapid LVO helps physicians speed up triage or transfer decision-making. Working in as few as 3 minutes, Rapid LVO uses a vessel tracker in conjunction with…
Read More