Archive for April 2017
Altus Spine: FDA Clearance of Titanium Interbody Fusion System
Altus Spine, based in West Chester, Pennsylvania, has announced the FDA clearance of the Silverstone Titanium Interbody Fusion (IBF) System, an implant meant to help stabilize and support the vertebrae during fusion. As indicated in the March 17, 2017 news release, “The Silverstone Titanium IBF System is a simple, yet comprehensive, system that includes both…
Read MoreNew WPI Center Hopes to Speed Up Medical Device Production
Worcester Polytechnic Institute is opening a new medical and technology development center it hopes will speed up the production of cutting edge medical devices and therapies for patients. PracticePoint at WPI, a public/private partnership announced Thursday, will include several testing suites to allow clinical researchers, engineers and manufacturers to more easily collaborate and commercialize robotic and…
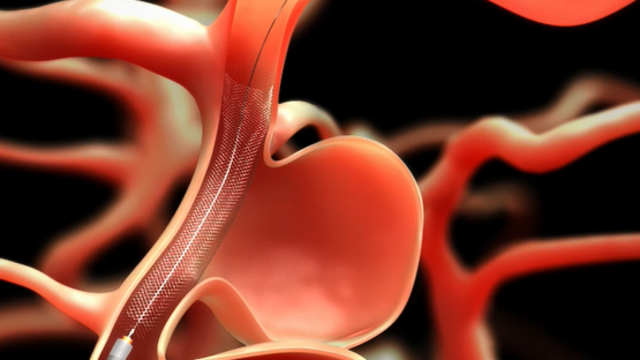
Read MoreDevice Slows Blood Flow into Brain Aneurysms
JACKSONVILLE, Fla. – A device that treats large brain aneurysms just as effective for treating smaller and sometimes harder to reach aneurysms, according to a study’s findings, which were presented at the International Stroke Conference Houston by Ricardo A. Hanel, MD, PhD, neurovascular surgeon with Baptist Health and Lyerly Neurosurgery and director of the Baptist Neurological Institute. One hundred and forty-one patients,…
Read MoreHospital Needle Pricks Easier with New Device
Scientists at Cincinnati Children’s Hospital and Ben Gurion University have developed a prototype device designed to quickly and accurately locate a vein or artery in children and adults in need of a medical procedure. It uses ultrasound and a robotic arm. FIND, or Fast Intelligent Needle Delivery, is the invention of the newly formed company,…
Read MoreRobotics Jobs in Medical Device – How do you know what search firms are “dialed in?” 5 Questions you should Ask:
There are only a handful of retained search firms that are leaders in surgical & medical robotics recruiting. We are putting together a list and will distribute in mid-May, 2017, but in the meantime… 5 Questions You should ask before sending your resume: Have you met in person with the executive management? When did you…
Read MoreAre Serious Medical-Device Hacks Imminent?
Regulators and medical-device-makers are bracing for an expected barrage of hacking attacks even as legal and technical uncertainties leave them in uncharted territory. Tens of millions of electronic health records have been compromised in recent years, a number that is growing and, some say, underreported. High-profile attacks have hit hospitals and health insurers, and now…
Read MoreInari Medical Announces First Patient Treated With The ClotTriever Thrombectomy System
IRVINE, Calif., April 10, 2017 /PRNewswire/ — Inari Medical Inc., a venture capital-backed medical device company focused on the interventional treatment of venous thrombus, announced the treatment of the first patient with its ClotTriever Thrombectomy System. The ClotTriever Thrombectomy System received 510(k) marketing clearance from the U.S. Food and Drug Administration on February 16 for the non-surgical…
Read MoreHealthcare’s Top VR Companies
Medical VR is an area with fascinating possibilities. It has not just moved the imagination of science-fiction fans, but also clinical researchers and real life medical practitioners. As a doctor, you could assist in the OR without ever lifting a scalpel. If you are a medical student, you could study the human body more closely and…
Read MoreInterventional cardiologists: Heroes of the witching hour
Coming off the ACC.17 conference, we have Margy Rockwood blogging for us about her experience: Last month, I attended the 2017 American College of Cardiology conference at the Walter E. Washington Convention Center in D.C., along with 13,000+ medical professionals and 6,000+ other attendees. Most of us made the trip for our annual infusion of…
Read More