Archive for October 2019
Smart Speaker Skill Monitors Baby’s Breathing Using White Noise
Researchers at the University of Washington (UW) have created a smart speaker skill that allows a device to track babies’ breathing and movement. BreathJunior plays white noise, recording it back to the speaker’s microphones to identify breathing motions. Researchers designed the system to be compatible with smart speakers such as Amazon Echo and Google Home.…
Read More3M Closes $6.7B Acelity Buyout – Acquisition Expands Presence in Advanced and Surgical Wound Care
3M announced it has completed the acquisition of Acelity, Inc. and its KCI subsidiaries worldwide from a consortium comprised of funds advised by Apax Partners (the Apax Funds), together with controlled affiliates of the Canada Pension Plan Investment Board (CPPIB) and the Public Sector Pension Investment Board (PSP Investments) for a total enterprise value of…
Read MoreLight Therapy Device to Treat Carbon Monoxide Poisoning
Carbon monoxide (CO)—which is produced by cars and trucks, as well as fires and explosions—is a toxic, colorless, and odorless gas, and there are more than 50,000 admissions to US emergency rooms due to CO poisoning each year, with many cases involving damaged airways and lungs. Investigators at Massachusetts General Hospital (MGH) recently developed a…
Read MoreMedTech of the Living Dead – How Scientists are Bringing Pig Brains Back to Life
Scientists at Yale University have developed a device that can restore circulation and cellular activity in a pig’s brain four hours after death, raising ethical questions about the possibilities of one day applying it to humans. BrainEx is an open-source device originally based on CaVESWave, a system developed by Biomed Innovations of North Carolina to preserve donated organs for…
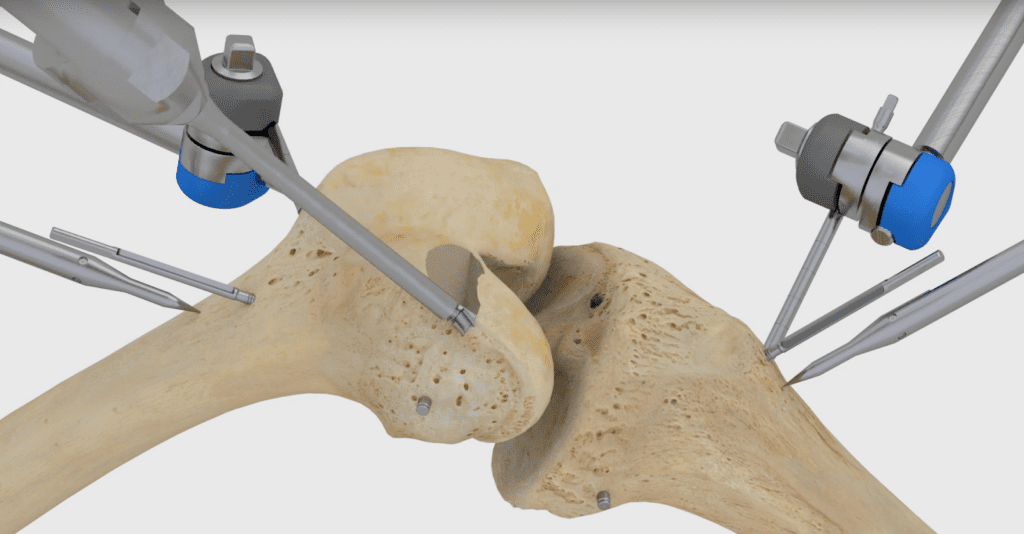
Read MoreTSolution Announces FDA Approval for a Robot Capable of Performing a Total Knee Replacement
Think Surgical Inc. this week announced that the U.S. Food and Drug Administration has granted clearance for it to market the TSolution One system for total knee replacement in the U.S. The robot already has CE Marking, and surgeons have used it in more than 550 procedures in the Asia-Pacific and European markets. “We are…
Read MoreMobile YuMi Robot Demos Future of Healthcare Robotics
ABB today opened its first global healthcare research hub on the Texas Medical Center (TMC) campus, in Houston, Texas. ABB showcased a number of concept technologies, including a mobile YuMi robot that assists with laboratory and logistics tasks in hospitals. The dual-arm, mobile YuMi will be able to sense and navigate its way around its…
Read MoreWrist Worn Step Trackers are as Accurate in Predicting Patient Health as Standardized Clinical Walking Tests
Determining how far patients with pulmonary disease can walk in six minutes has long been an effective clinical tool to help physicians determine their exercise capacity, as well as to aid in predicting health outcomes and mortality. Now, in a new study, researchers at Intermountain Healthcare in Salt Lake City found that steps measured through…
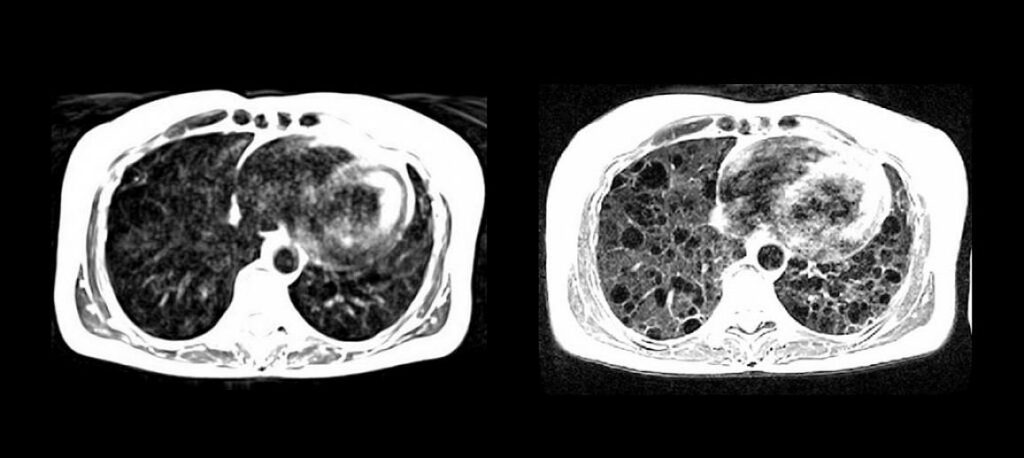
Read MoreNIH Researchers Develop MRI with Lower Magnetic Field for Cardiac and Lung Imaging
National Institutes of Health researchers, along with researchers at Siemens, have developed a high-performance, low magnetic-field MRI system that vastly improves image quality of the lungs and other internal structures of the human body. The new system is more compatible with interventional devices that could greatly enhance image-guided procedures that diagnose and treat disease, and…
Read MoreAngioDynamics Acquires Eximo Medical, Ltd. and its Innovative 355nm Laser Atherectomy Technology
AngioDynamics, Inc., a leading provider of innovative, minimally invasive medical devices for vascular access, peripheral vascular disease, and oncology, today announced that it has acquired Eximo Medical, Ltd., an early commercial stage, medical device company, and its proprietary 355nm wavelength laser-technology platform for $46 million in up-front consideration with up to $20 million of contingent…
Read MoreAbbott and Edwards Lifesciences May Have Mislabeled Hundreds of Patient Deaths
Abbott and Edwards Lifesciences may have mislabeled patient deaths as injuries or device malfunctions in hundreds of FDA adverse event reports involving their transcatheter aortic or mitral valve replacement devices, according to a new study. The study by JAMA Internal Medicine editor Dr. Rita Redberg, former FDA unique device identification (UDI) external program manager Madris Tomes and others examined a total of…
Read More