Biologics, Wound Care, Infection Prevention
New Study Finds HealthySole UVC Technology Deactivates COVID-19 on Footwear
Researchers have proven a device using ultraviolet light technology can neutralize the COVID-19 virus and other infectious diseases on the soles of shoes by more than 99.5 percent, according to a new study. The device, called HealthySole® PLUS, is being introduced in hospitals and other settings where infection control is urgent. A home version of the…
Read MoreAdvanced Sterilization Products Granted FDA EUA Allowing Decontamination of Millions of Compatible N95 Respirators to Help Protect Against COVID-19
Advanced Sterilization Products (ASP), a Fortive (NYSE:FTV) company and global leader in infection prevention, has received Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) for the use of STERRAD® Systems to decontaminate compatible N95 respirators.1 Utilizing equipment already available onsite in many US hospitals, STERRAD Systems could collectively reprocess millions of compatible N95 respirators daily.…
Read MoreVomaris Seeking Expedited FDA Approval for New Virus-Killing Face Mask
Vomaris Innovations, Inc., headquartered in Tempe, AZ, announced today that significant research findings confirmed viruses are killed upon exposure to the company’s bioelectric V.Dox™ Technology platform. Vomaris has begun applying their technology to manufacture prototype face masks that can be worn alone or inside an existing face mask with the intention of killing viruses and reducing…
Read MoreCelularity Announces FDA Clearance of IND Application for CYNK-001 in Coronavirus, First in Cellular Therapy
Celularity Inc., a clinical-stage company developing allogeneic cellular therapies from human placentas, today announced the U.S. Food and Drug Administration (FDA) has cleared the Company’s Investigational New Drug (IND) application for the use of its proprietary CYNK-001 in adults with COVID-19. With this, Celularity will commence a Phase I/II clinical study including up to 86…
Read MoreBattelle Announces Rapid Manufacturing of a System to Decontaminate N95 Respirator Masks
Battelle announced today that it has begun rapid manufacturing of a system to decontaminate N95 respirator masks and other medical protective equipment. Each Battelle CCDS Critical Care Decontamination System™ is capable of decontaminating up to 80,000 masks per day at full capacity. Because it is scalable, the system is capable of processing even more pieces…
Read MoreNorthwell Implements New On-site Laboratory Treatment Technology for COVID-19 Infected Medical Waste
To minimize the risk of spreading COVID-19, Northwell Health has become the first US health system to implement a new technology that breaks down medical waste from laboratory testing in a safe, environmentally friendly way. Developed by Irish-based Technopath Clinical Diagnostics, the Envetec 200 system simultaneously shreds and disinfects infectious waste using a patented destruction and disinfection…
Read MoreBiostage Announces IND Approval from FDA for its Lead Product Candidate Cellspan Esophageal Implant
Biostage, Inc., a bioengineering company developing next-generation esophageal implants, today announced that the U.S. Food and Drug Administration (FDA) has approved the Company’s Investigational New Drug application (IND) for the Cellspan Esophageal Implant (CEI) to treat patients with end-stage esophageal disease that require a segmental surgical resection to repair the diseased tissue. The FDA notified…
Read MoreBIOLASE Announces Regulatory Clearance of Laser Bacterial Reduction Therapy Indication for Epic Hygiene Laser
BIOLASE, Inc., the global leader in dental lasers, is pleased to announce the Epic Hygiene™ laser received regulatory clearance for Laser Bacterial Reduction (LBR) therapy indication from the Food and Drug Administration (FDA). This new indication now allows for the early management of periodontal disease utilizing laser light energy to reduce bacteria and thus decreasing…
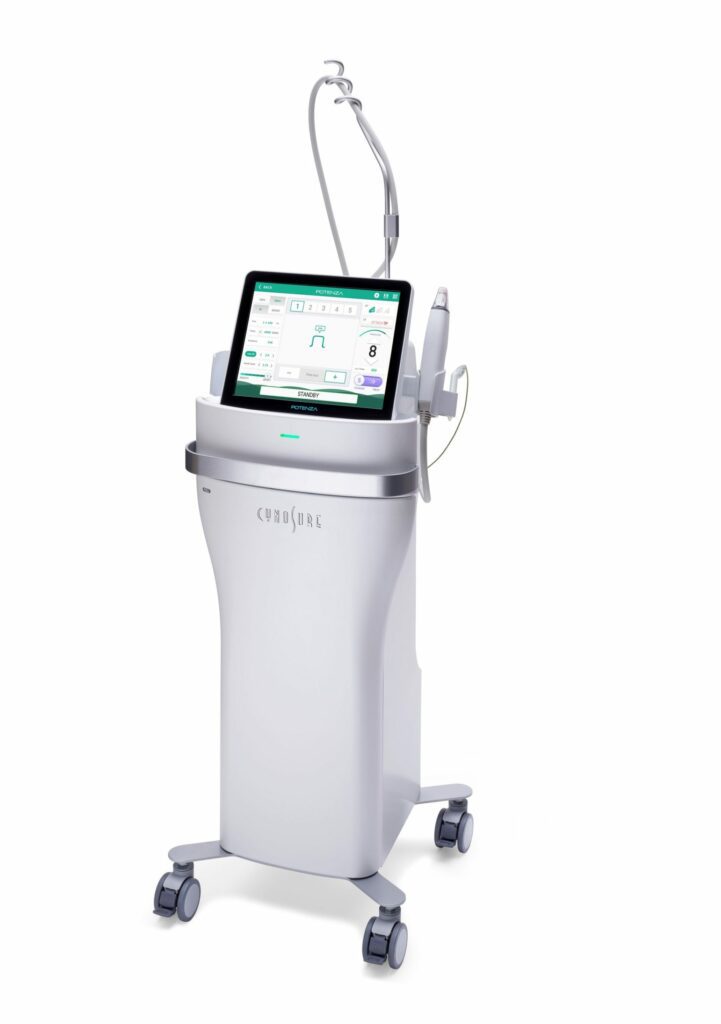
Read MoreCynosure Launches Potenza Radiofrequency Microneedling Device Expanding Company’s Growing Skin Revitalization Portfolio
Cynosure, the global leader in medical aesthetics, announced today the U.S. Food and Drug Administration (FDA) clearance of the Potenza™ radiofrequency (RF) microneedling device, the first and only FDA-cleared four-mode RF microneedling device offering clinicians unrivaled versatility and personalized treatments for patients. The new standard in RF microneedling, the Potenza device’s four modes (monopolar or…
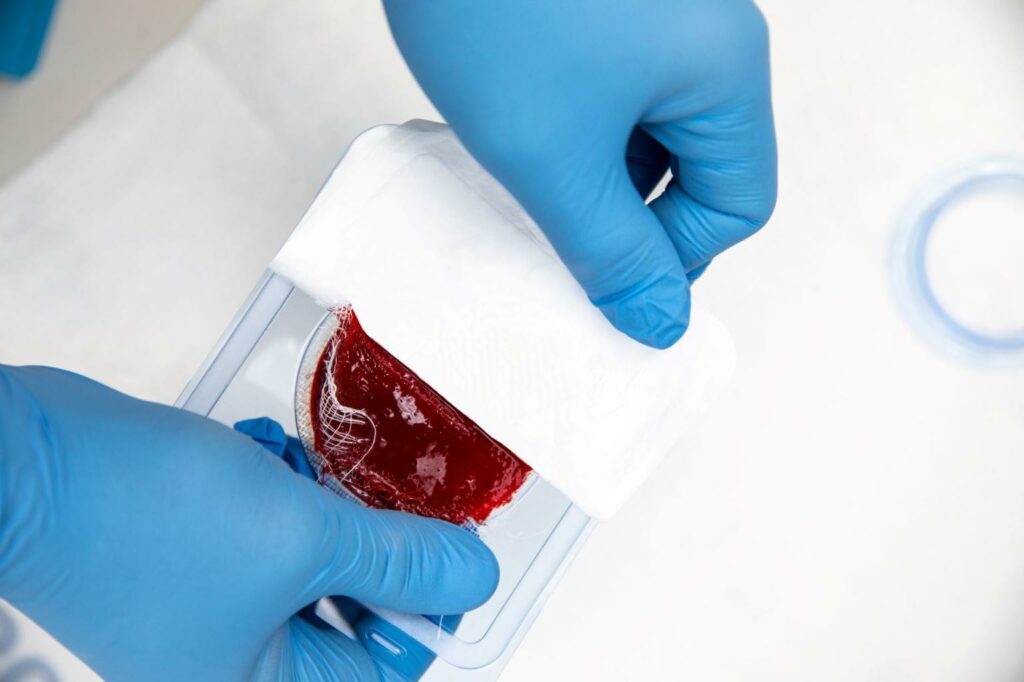
Read MoreRedDress Announces Launch of ActiGraft, FDA-Cleared for the Treatment of Chronic and Acute Wound Types
RedDress, a privately held, Israel and U.S. based company, today announced the U.S. launch of ActiGraft, the first wound treatment that transforms – in real time – a patient’s blood into an autologous whole blood clot tissue. Once applied, ActiGraft serves as a protective covering, biologic scaffold and wound microenvironment to promote the natural wound healing processes…
Read More